Clinical Research and Toxicological assessment of Clindamycin phosphate
Clindamycin phosphate is an antibiotic that works by stopping the growth of bacteria, helping to reduce the number of acne lesions. Once introduced into the body, clindamycin phosphate is hydrolyzed to form clindamycin, which possesses antibacterial activity. It has significant antibacterial effects against Gram-positive bacteria such as Staphylococcus aureus and Streptococcus species, as well as against anaerobes including Bacteroides and Clostridium species. Importantly, it exhibits no cross-allergenicity with penicillin or cephalosporin antibiotics, making it suitable for use in patients allergic to penicillin.

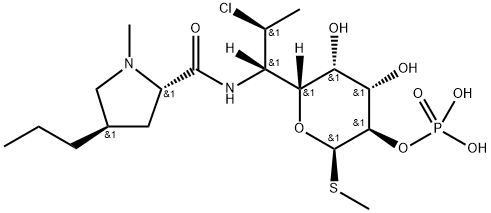
Figure1: Picture of Clindamycin phosphate
Basic Introduction
Clindamycin phosphate is a widely used antibiotic with specific utility in the therapy of pulmonary and intra-abdominal anaerobic infections. The recent emphasis on cost containment has encouraged strategies such as reducing the frequency of drug administrations in an effort to control therapy costs. In the case of clindamycin phosphate, this has led to a wide range of recommended regimens for the treatment of serious intra-abdominal infections. Many of these regimens, however, have not been subjected to rigorous clinical evaluation. As a result, the preferred regimen at many institutions, based on comparative clinical studies, remains the administration of clindamycin phosphate at a dose of 600 mg every six hours, combined with adequate coverage of gram-negative bacteria. The pharmacokinetic profile of clindamycin phosphate is complex because the compound itself is administered intravenously in its ester form. Following infusion, clindamycin phosphate is converted into the active moiety, clindamycin, which may therefore be considered a metabolic product of clindamycin phosphate. [1]
Pharmacokinetics
A parenteral formulation of clindamycin is necessary for the treatment of serious infections or when oral therapy is contraindicated. Clindamycin phosphate, the 2-phosphate ester of clindamycin, is distinct from clindamycin hydrochloride or palmitate hydrochloride in that it is specifically designed for parenteral administration. Although clindamycin phosphate is inactive in vitro, it serves as a prodrug that, after intravenous or intramuscular injection, is rapidly hydrolyzed to form microbiologically active clindamycin. Following intramuscular injection, clindamycin phosphate is well absorbed, with peak serum concentrations of the active drug achieved within three hours in adults and within one hour in children. An intramuscular dose of 300 mg of clindamycin phosphate typically results in an average peak serum concentration of 5 µg/mL. After intravenous administration of clindamycin phosphate, the serum half-life of the resulting active clindamycin is approximately three hours in adults and two and a half hours in children. The active clindamycin derived from clindamycin phosphate is highly effective against Staphylococcus aureus and streptococci (except Streptococcus faecalis), as well as against most anaerobic bacteria that cause infections in humans. [2]
Clinical Research
Clindamycin phosphate has been formulated as an aqueous gel containing 1.2% solubilized clindamycin phosphate in combination with 0.025% tretinoin, which is present in both solubilized and crystalline forms. This clindamycin phosphate /tretinoin combination gel was evaluated in three pivotal phase 3 studies, as well as in other investigations including a 52-week trial. Across the pivotal studies, a total of 4,550 participants aged 12 years and older with mild, moderate, or severe acne vulgaris were enrolled. In these trials, clindamycin phosphate, as part of the combination gel, contributed to a significant reduction in both inflammatory and noninflammatory lesions. The formulation containing clindamycin phosphate and tretinoin was well tolerated and proved superior to either active ingredient alone and to the vehicle, regardless of acne severity. This article discusses the vehicle characteristics of the gel containing clindamycin phosphate that appear clinically relevant, its stability profile—including results after exposure to UV light and/or benzoyl peroxide—and its cutaneous tolerability. [2]
Toxicological assessment
In the present study, three patients (3%) developed rashes. Clindamycin phosphate was administered to the first patient for 13 days, after which the rash appeared upon switching to oral clindamycin. In the second patient, the rash occurred 48 hours after initiating clindamycin phosphate therapy, following a prior course of ampicillin. The third patient developed a rash while on oral clindamycin after completing a four-day course of clindamycin phosphate. In the latter two cases, clindamycin phosphate had been transitioned to oral therapy, and the patients continued oral clindamycin despite the rash without any adverse effects. However, in the first case, clindamycin was discontinued. The rash was maculopapular and itchy, identical to that described in patients receiving oral clindamycin, in whom the incidence can reach up to 10%. Additionally, diarrhea developed in two patients after two and nine days of clindamycin phosphate therapy, respectively. In both cases, clindamycin phosphate was discontinued, and the patients recovered within 24 hours. [3]
Reference
[1] Plaisance K I, Drusano G L, Forrest A, et al. Pharmacokinetic evaluation of two dosage regimens of clindamycin phosphate[J]. Antimicrobial agents and chemotherapy, 1989, 33(5): 618-620.
[2] Del Rosso J Q, Jitpraphai W, Bhambri S, et al. Clindamycin phosphate 1.2%-tretinoin 0.025% gel: vehicle characteristics, stability, and tolerability[J]. Cutis, 2008, 81(5): 405-408.
[3] Finch R G, Phillips R G, Geddes A M. A clinical, microbiological and toxicological assessment of clindamycin phosphate[J]. Journal of Antimicrobial Chemotherapy, 1975, 1(3): 297-303.
You may like
Related articles And Qustion
Lastest Price from Clindamycin phosphate manufacturers

US $1.00/g2025-12-15
- CAS:
- 24729-96-2
- Min. Order:
- 300g
- Purity:
- 99.8%
- Supply Ability:
- 20 TONS

US $0.00-0.00/kg2025-12-09
- CAS:
- 24729-96-2
- Min. Order:
- 1kg
- Purity:
- 99%
- Supply Ability:
- 100kg