Clinical Study and Therapeutic Use of Indobufen
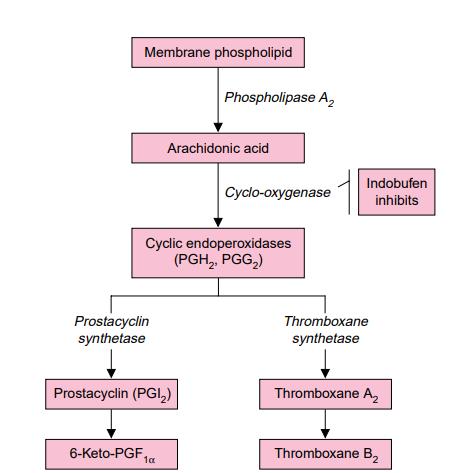
Indobufen exerts its antiplatelet effect by reversibly inhibiting the enzyme cyclooxygenase in platelets, which leads to a reduction in thromboxane synthesis. The efficacy of oral indobufen has been investigated in clinical trials for several indications: the secondary prevention of thromboembolic complications in patients with or without atrial fibrillation, the prevention of graft occlusion following coronary artery bypass graft (CABG) surgery, and the treatment of intermittent claudication.

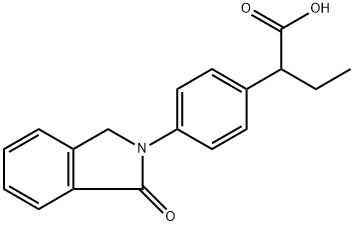
Figure1: Picture of Indobufen
Overview
Indobufen, chemically known as (±)-2-[p-(1-oxo-2-isoindolinyl)phenyl]butyric acid, is an orally active antiplatelet agent. It inhibits key platelet functions, including activation, adhesion, and aggregation. These effects are primarily achieved because Indobufen induces a reversible inhibition of arachidonic acid metabolism at the level of the platelet cyclo-oxygenase enzyme. Unlike aspirin, which causes an irreversible acetylation of cyclo-oxygenase, the antiplatelet action of Indobufen is short-lived. Following the cessation of administration, normal platelet function typically resumes within 24 hours. Consequently, Indobufen is associated with a reduced risk of causing the prolonged bleeding time that is generally seen with other antiplatelet therapies. The therapeutic efficacy of Indobufen in treating cerebral, peripheral, and coronary vascular diseases has been documented in earlier literature. This review provides an update on data concerning its use, specifically in preventing graft occlusion after arterial bypass surgery and in treating patients with intermittent claudication. However, the primary focus here is on the role of Indobufen in the prophylaxis of thromboembolic events in high-risk patients, particularly those with nonrheumatic atrial fibrillation. [1]
Clinical Study
Clinical trial data support the efficacy of indobufen in the secondary prevention of thromboembolic events. In a one-year, non-blind study, indobufen administered at 200mg once or twice daily demonstrated significantly greater effectiveness compared to no treatment, although its effect was less pronounced than that of ticlopidine (250mg once or twice daily). Additionally, a smaller 28-month trial involving patients at elevated risk for systemic embolism (half of whom had atrial fibrillation) showed that indobufen (100mg twice daily) significantly reduced the risk of stroke relative to placebo. Furthermore, the one-year Italian Atrial Fibrillation Study (SIFA) specifically evaluated patients with non-rheumatic atrial fibrillation who had recently experienced a cerebrovascular event. In this trial, a regimen of indobufen (100mg or 200mg twice daily) proved equally effective as dose-adjusted warfarin (targeted to an International Normalised Ratio of 2.0 to 3.5) for preventing recurrent thromboembolic events. The incidence rates for the composite endpoint of major vascular events (10.6% vs. 9.0%) and for recurrent stroke (5% vs. 4%) were comparable between the indobufen and warfarin treatment groups. [1]
Pharmacodynamic Properties
Indobufen is a derivative of isoindolinyl phenyl-butyric acid. It reversibly inhibits platelet aggregation in vitro and ex vivo by targeting the enzyme platelet cyclo-oxygenase, which leads to the suppression of thromboxane A2 production—a potent platelet activator. The ex vivo suppression of platelet thromboxane synthesis by Indobufen becomes evident within 2 hours of administration (98%), remains significant at 12 hours (89%), and is attenuated at 24 hours (47%), indicating that its inhibitory action is reversible. In platelets obtained from both healthy volunteers and patients with ischemic heart disease, atherosclerosis, or angina pectoris, Indobufen inhibits the release reaction and the secondary wave of aggregation induced by platelet agonists such as adenosine diphosphate, adrenaline (epinephrine), and platelet activating factor. Following a single oral dose of 200mg, the maximum inhibitory effect of Indobufen on agonist-induced platelet aggregation is observed at 2 hours, and this inhibition is reversible, with platelet aggregation returning to baseline values within 24 hours. Indobufen alone or in combination with pentoxifylline reduces the levels of β-thromboglobulin and platelet factor 4, which are markers of platelet activation. Additionally, Indobufen has other effects, including decreased platelet adhesiveness and improved red blood cell deformability. It prolongs bleeding time, although this remains within the upper limit of normal and for a shorter duration than aspirin (acetylsalicylic acid) or ticlopidine. Indobufen also improves microcirculatory parameters such as skin blood flow and the venoarteriolar reflex in patients with intermittent claudication. Moreover, Indobufen has been shown to inhibit thrombus formation in animals and prevent graft occlusion following vascular surgery. [1]
Pharmacokinetic Properties
Indobufen demonstrates rapid and complete absorption following oral administration, achieving peak plasma concentrations within 2 hours. The drug exhibits an elimination half-life (t½β) of approximately 7 hours. Indobufen is characterized by a low apparent volume of distribution (approximately 15L), which is a direct consequence of its very high plasma protein binding (>99%). Within 48 hours post-administration, Indobufen leads to the excretion of 70% to 80% of the administered dose in urine. The majority is eliminated renally as glucuronic acid conjugates, while 11% to 13% is excreted unchanged. The excretion profile of Indobufen is not influenced by its route of administration (oral or intravenous), and its renal clearance remains unaffected by the presence of food. However, Indobufen requires careful dosage consideration in the elderly and in patients with renal insufficiency, as its clearance is significantly prolonged in these populations. Regarding drug interactions, few have been reported for Indobufen. A documented interaction includes an increase in the area under the concentration-time curve (AUC) of the concomitant drug glipizide. [1]
Therapeutic Use
Indobufen, at a dosage of 200mg twice daily, significantly improves walking distances in patients with intermittent claudication. Compared to placebo, Indobufen significantly increased walking distances within the first 10 days of therapy, with continued improvement observed over a six-month treatment period. In a nonrandomised comparative study, Indobufen 200mg twice daily demonstrated a more pronounced improvement in both pain-free and total walking distance than aspirin 500mg twice daily after one year. The efficacy of Indobufen has also been evaluated in maintaining graft patency following surgery. At a dosage of 200mg twice daily, Indobufen was as effective as a combination of aspirin and dipyridamole in reducing the incidence of reocclusion after coronary artery bypass graft surgery, with both treatment groups showing a 34% frequency after one year. Furthermore, Indobufen reduced platelet accumulation and lesion formation in patients following carotid endarterectomy when compared to placebo over a six-month period. In the context of secondary prevention, preliminary investigations suggest Indobufen may provide prophylactic benefits for patients following a transient ischemic attack, mild stroke, or acute myocardial infarction. In one study, the incidence of secondary cardiovascular events was significantly lower in patients treated with Indobufen 100mg twice daily compared to placebo over 26 months. An initial study also indicates that Indobufen may have a prophylactic effect in patients with classic or common migraine. Finally, Indobufen has been studied for its effect on hemodialysis efficiency. Clinical trials have shown that Indobufen significantly reduces platelet aggregation on hemodialysis membranes. Administration of Indobufen 200mg twice daily for seven days resulted in a lower rate of membrane occlusion compared to placebo, suggesting that Indobufen therapy can improve the efficiency of hemodialysis. [2]
Place of Indohufen in Therapy
Indobufen is a platelet aggregation inhibitor with therapeutic potential in thrombosis management. Studies have shown that Indobufen is an effective antiplatelet agent which, in contrast to aspirin, causes reversible inhibition of platelet cyclo-oxygenase without exerting clinically significant effects on haemodynamic parameters. The majority of clinical investigations to date have focused on the therapeutic efficacy of Indobufen in patients with peripheral vascular disease and on its prophylactic use following revascularization surgery. Placebo-controlled, randomised studies have established that Indobufen therapy leads to a significant and sustained increase in both pain-free and total walking distances in patients with intermittent claudication. Preliminary findings from a nonrandomised, short-term study suggest that Indobufen may have a more pronounced therapeutic effect than aspirin in this patient population; however, this observation requires confirmation through well-controlled studies. Furthermore, the comparative efficacy of Indobufen versus ticlopidine in peripheral artery disease remains to be definitively established. The clinical relevance of the apparent beneficial effect of Indobufen on microcirculatory parameters, as observed in studies of patients with peripheral vascular disease or diabetes, has yet to be determined. When administered at a dosage of 200mg twice daily, Indobufen appears to be significantly more effective than placebo in preventing vascular reocclusion following carotid endarterectomy and femoropopliteal bypass surgery. In the context of coronary artery bypass graft patency, Indobufen is at least as effective as a combination regimen of aspirin and dipyridamole. Beyond its established roles, the efficacy of Indobufen for the secondary prevention of vascular events following myocardial infarction or transient ischemic attack has been explored in a limited number of controlled studies. While these investigations suggest a potential benefit for Indobufen in reducing the incidence of secondary events over a three-year period, further long-term studies are necessary to confirm its prophylactic efficacy and its influence on patient prognosis. [2]
Reference
[1] Bhana N, McClellan K J. Indobufen: an updated review of its use in the management of atherothrombosis[J]. Drugs & aging, 2001, 18: 369-388.
[2] Wiseman L R, Fitton A, Buckley M M. Indobufen: a review of its pharmacodynamic and pharmacokinetic properties, and therapeutic efficacy in cerebral, peripheral and coronary vascular disease[J]. Drugs, 1992, 44(3): 445-464.
You may like
Related articles And Qustion
See also
Lastest Price from Indobufen manufacturers

US $0.00-0.00/kg2025-05-21
- CAS:
- 63610-08-2
- Min. Order:
- 1kg
- Purity:
- 99% up by HPLC
- Supply Ability:
- 20tons

US $0.00-0.00/Kg/Drum2025-04-21
- CAS:
- 63610-08-2
- Min. Order:
- 1KG
- Purity:
- 99%minHPLC
- Supply Ability:
- 1000KG