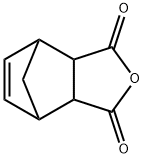
Copolymerization and Spectroscopy of Nadic Anhydride
Nadic anhydride is produced via the Diels-Alder reaction between cyclopentadiene and maleic anhydride, with the endo configuration predominating in industrial applications. It serves as a crucial high-temperature curing agent for epoxy resins, offering excellent weather resistance, electrical insulation, and mechanical properties. Additionally, it enhances the air-drying properties, heat resistance, and corrosion resistance of unsaturated polyester resins. Furthermore, it functions as a resin modifier, rubber intermediate, surfactant, and insecticide raw material, finding extensive applications in electronics, aerospace, coatings, and other sectors.

Copolymerization of nadic anhydride with styrene
Polymer properties can be greatly improved by the process of copolymerization and various copolymer materials excessively used in diverse applications. The copolymer properties can be tuned by changing the monomers, composition of monomers, and copolymerization techniques. Reactivity ratios can offer information such as copolymer composition, relative reactivity of monomer pairs, etc. We used nadic anhydride as it contains an isolated unsaturation quite away from the influence of the carbonyl or anhydride group. In other words, the reactivity of the unsaturation in nadic group in its anhydride is not affected by the other groups. If at all, it can experience a minor inductive effect of the distant anhydride group only. Therefore, we chose nadic anhydride for this study, as this facilitates the estimation of the copolymer composition through a simple acid–base titration. The anhydride, unlike the acid or imide, has solubility in many aprotic solvents. Ryan Baumgartner et al explored the alternating ring opening polymerization between nadic anhydride and various epoxides to form polyesters. Maleic anhydride-modified polypropylene is the utmost significant, commercially functionalized PP polymer for its low cost, high activity, and good processability. Many reports are available on the free radical copolymerization of the nadic analogue like maleic anhydride or its derivatives. However, no information on the copolymerization of nadic–styrene pair is available in the open literature. Hence, we undertook a study to generate data on the reactivity ratios of this pair.[1]
In normal course on such cases, the allylic protons of the monomers transfer protons to their allylic position as allylic-free radicals are stabilized by way of conjugation with the adjacent π electrons; this demands a coplanarity for the allylic radical. In this case, although allylic protons are there, they are located on a bridge head so the coplanarity requirement is blocked. The propensity for polymerization is reduced by the presence of nadic anhydride, decreases the rate of copolymerization of styrene; however, this trend is levelled off at a nadic anhydride mole fraction of 0.5 and beyond. We successfully synthesized nadic anhydride-styrene copolymer system and determined the reactivity ratio. Reactivity ratios were calculated by using three different methods, Fineman–Ross, Kelen–Tüdös, and extended Kelen–Tüdös. They form an azeotropic pair at a feed composition of 0.195 mole fraction for nadic anhydride (NA). This reactivity ratio helps to calculate the sequencing of the functional monomer NA in the copolymer. In the copolymer formed, the anhydride group can be made use of for mounting other molecules making use of its high electrophilic nature. Nadic group's ready polymerizability is explained based on difficulty in free radical transfer to the allylic position due to geometric restrictions, though it retards the overall copolymerization rate.
The vibrational spectra of norbornene and nadic anhydride
Thermosetting resins are used in high temperature applications such as resin–carbon fibre composites for aerospace components. The thermoset resins are highly cross-linked, insoluble, intractable materials. In particular, the nature of the cross-links present is still uncertain.2Vibrational spectroscopy has played a key role in understanding the chemistry of these materials in the early stages of the cure process and potentially can offer insights at later stages. Inelastic neutron scattering (INS) spectroscopy is particularly attractive here, because it allows the bulk of the material to be studied, even in the presence of carbon or silica fibres. One such derivative is nadic anhydride (cis-endo-5-norbornene-2,3-dicarboxylic anhydride) which is used either directly or in the form of its methyl half-ester. The goal of this paper is to complete the vibrational assignment of norbornene and assign that of nadic anhydride. This is with the long-term aim of being better able to understand the chemistry, and in particular the cross-links, in thermosetting polyimides. To this end we have obtained the solid state inelastic neutron scattering (INS), infrared and Raman spectra.[2]
This paper demonstrates that with state-of-the-art INSspectra it is possible to go beyond this and successfully analyse complex systems that have not been studied before. This approach has allowed the assignment of norbornene to be completed and has enabled a related but more complex molecule to be assigned for the first time. This is noteworthy because nadic anhydride is a low symmetry molecule of medium size, it is unusual to attempt the analysis of such a system without a library of related compounds and the use of isotopic substitution. INS data is essential in this case because it provides confidence in the ab initio results. The installation of TOSCA at ISIS has resulted in an almost twofold improvement in resolution over previous instruments and is the best available anywhere in the world. The quality of the data that is obtainable is quite remarkable. It is apparent that for the first time, INS data of the same quality, in terms of both resolution and signal-to-noise ratio, as infrared and Raman spectra is routinely available. It is accepted that the comparison is not fair; the spectra were recorded at different temperatures, from different amounts of sample and with different measurement times. Nonetheless, the basic point about the quality of data is valid.
References
[1] P. A. Jeemol, C. N., S. Mathew. (2021). Copolymerization of nadic anhydride with styrene: Reactivity ratios. Polymers for Advanced Technologies, 32 1, 1888–1894.
[2] Parker, S. F., Williams, K. P. J., Steele, D., & Herman, H. (2003). The vibrational spectra of norbornene and nadic anhydride†. Physical Chemistry Chemical Physics, 8, 1508–1514.
See also
Lastest Price from Nadic anhydride manufacturers
US $1.00/kg2025-04-21
- CAS:
- 826-62-0
- Min. Order:
- 1kg
- Purity:
- 99%
- Supply Ability:
- 10 mt

US $10.00/kg2025-04-21
- CAS:
- 826-62-0
- Min. Order:
- 1kg
- Purity:
- 99%
- Supply Ability:
- 20 ton