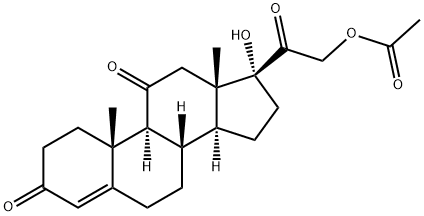
Cortisone Acetate: Clinical Use and Metabolic Comparison
Cortisone acetate is used to treat a variety of inflammatory, autoimmune, and hormonal conditions. It works by suppressing the body’s immune response and reducing inflammation. It also replaces low levels of steroid hormones in people with reduced adrenal gland function. Common side effects include fluid retention, high blood sugar, increased blood pressure, behavior and mood changes, increased appetite, and weight gain. Cortisone acetate was first isolate in 1935 and became more widely researched in 1949. Since then, glucocorticoids have been used to treat a number of inflammatory conditions such as endocrine, rheumatic, collagen, dermatologic, allergic, ophthalmic, respiratory, hematologic, neoplastic, edematous, and gastrointestinal diseases and disorders.

Therapy shift from cortisone acetate to modified-release hydrocortisone
Adrenal insufficiency (AI) is characterized by insufficient cortisol production; this can be caused by either primary adrenal failure (PAI - Addison’s disease) or inadequate secretion of ACTH from the pituitary gland. In the absence of prompt replacement therapy, this condition is potentially lethal, thus treatment with glucocorticoids (GC) is essential. This treatment aims to reproduce the physiological circadian pattern of cortisol secretion with higher levels in the morning that decrease during the day reaching the nadir at night, with adjustments during periods of stress. However, classical replacement therapy with cortisone acetate (CA) or hydrocortisone (HC), administered with two or three daily doses, has been demonstrated to expose patients to supraphysiological cortisol levels. Side effects of cortisol excess are mainly represented by glycometabolic alterations, cardiovascular disease, hypertension, obesity, osteoporosis, risk of infections, and, consequently, a reduction of QoL and increased mortality. The primary aim of our study was to evaluate in our cohort of patients who shifted from Cortisone acetate (the most used substitutive glucocorticoid therapy in Italy) to MRH, the long-term effects on glucose metabolism, blood pressure, bone health, and subjective disease control. The secondary aim was to assess the effective total daily exposure to GCs, using salivary cortisol.[1]
Data on glycometabolic, cardiovascular, and bone health assessment after shifting to MRH were compared to patients continuing Cortisone acetate. Over a median follow-up of 35 months, we did not observe any significant modification in all the examined parameters in both groups except for arterial hypertension control and SDC-score, which improved more frequently in patients who shifted to MRH. The analyses of the salivary cortisol curve showed that daily cortisol profile with MRH treatment was more similar to healthy subjects than CA-group. However, in both MRH and CA groups, the overall cortisol daily exposure was higher than controls, especially in the first part of the day with MRH and in the second part with the Cortisone acetate. Quality of life questionnaires specifically tailored for AI patients often appear hardly evaluable in clinical practice. Regarding metabolic parameters, we only assessed glycemic control, and no data about lipids were analyzed. However, this is the first study comparing CA with MRH, suggesting the need for a tailored therapy shift due to the different pharmacokinetics of the two drugs. In conclusion, in our experience, we found a globally positive role of MRH in AI treatment, especially in blood pressure control and subjective disease control. The lack of amelioration in glucose metabolism and total cortisol daily exposure could suggest the need for a dose reduction when shifting from Cortisone acetate to MRH. More studies, with a longer follow-up and larger cohorts, are needed to confirm possible positive effects on bone health.
Metabolic Effects of Cortisone Acetate vs Hydrocortisone
Approximately 75% of European patients with adrenal insufficiency (AI) receive hydrocortisone for glucocorticoid replacement. In some countries, however, cortisone acetate (CA) is more commonly used. The pharmacokinetic profiles of HC and CA differ. HC is the chemical equivalent of endogenous cortisol and, thus, is a biologically active GC, whereas CA requires conversion to cortisol by the enzyme 11-β-hydroxysteroid dehydrogenase type 1 to become active. Therefore, orally administered CA has a delayed peak of serum cortisol concentration compared to HC. In a previous study, the use of Cortisone acetate was associated with more favorable glucose metabolism, with glycated hemoglobin (HbA1c) levels being lower than in patients treated with HC. Thus, HC may have a more potent metabolic effect than the assumed equivalent dose of CA. In Sweden, Cortisone acetate was the conventional GC used for replacement therapy in patients with AI until 2001, when it was withdrawn from the market by the manufacturer and HC was approved by the Swedish Medical Products Agency. We were therefore able, in an ongoing prospective study, to follow patients with hypopituitarism through a period during which most patients were switched from CA to HC. The aim of this study was to assess the consequent metabolic effects of the change in GC replacement in patients with SAI.[2]
In this study we evaluated whether the type of GC for replacement therapy has an impact on metabolic outcome in patients with SAI. We found that a switch from Cortisone acetate to an assumed equivalent dose of HC was associated with an increase in body weight, total fat mass, waist circumference, and HbA1c. These data suggest that the metabolic potency of HC in relation to CA is greater than previously thought when using the conversion ratio of 4:5 for HC:CA. In conclusion, our findings suggest that the type of GC, and not only the dose, has affects the metabolic profile in patients with SAI. In this study, the switch from CA to an assumed equivalent dose of HC led to adverse metabolic effects, suggesting that the widely used conversion ratio of 4:5 for HC to Cortisone acetate may be imprecise and underestimate the metabolic potency of HC. We can also speculate that, in patients with SAI, replacement treatment with CA may have more favorable pharmacokinetic properties in terms of metabolic outcomes.
References
[1]Frigerio S, Carosi G, Ferrante E, Sala E, Polledri E, Fustinoni S, Ambrosi B, Chiodini I, Mantovani G, Morelli V, Arosio M. Effects of the therapy shift from cortisone acetate to modified-release hydrocortisone in a group of patients with adrenal insufficiency. Front Endocrinol (Lausanne). 2023 Jan 24;14:1093838. doi: 10.3389/fendo.2023.1093838. PMID: 36761196; PMCID: PMC9902698.
[2]Ekstrand E, Esposito D, Ragnarsson O, Isgaard J, Johannsson G. Metabolic Effects of Cortisone Acetate vs Hydrocortisone in Patients With Secondary Adrenal Insufficiency. J Endocr Soc. 2020 Oct 29;4(12):bvaa160. doi: 10.1210/jendso/bvaa160. PMID: 33241171; PMCID: PMC7671249.
You may like
See also
Lastest Price from Cortisone acetate manufacturers
US $0.00-0.00/kg2025-05-20
- CAS:
- 50-04-4
- Min. Order:
- 1kg
- Purity:
- 98%min
- Supply Ability:
- 100kg

US $10.00/KG2025-04-21
- CAS:
- 50-04-4
- Min. Order:
- 1KG
- Purity:
- 99%
- Supply Ability:
- 10 mt