Cyclopropanecarboxaldehyde: Conformational Isomerization & Astrochemical Significance
Cyclopropanecarboxaldehyde is characterized by its cyclopropane ring and aldehyde functional group, making it a valuable intermediate in various chemical syntheses. This substance reacts with Grignard or organolithium reagents to give the expected secondary alcohols.

Solid-State Isomerization of Isomers of Cyclopropanecarboxaldehyde
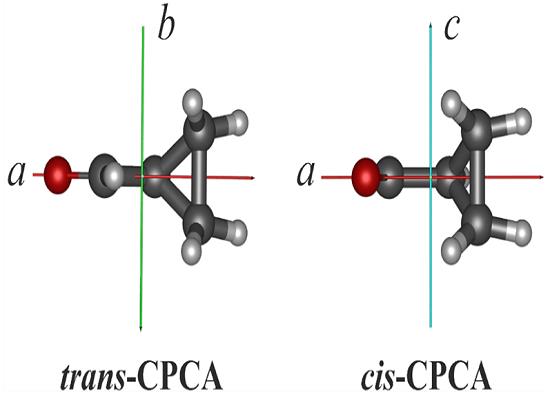
Over 200 molecules and molecular ions have been reported to be present in the gas phase of the interstellar medium (ISM), and to these can be added about six firm spectroscopic identifications in interstellar ices, and about six other solid-phase identifications that are more tentative. With all of these in hand it is possible to search for trends and to identify molecular families from which to make predictions of as yet undiscovered species. In this paper, we consider one such molecular family and use it as motivation for new solid-phase measurements on one of its members and two related compounds. While there is an extensive astrochemical literature on aromatic rings, far less information is available related to aliphatic rings. Here shows the smallest saturated carbon ring, the prototypical cyclopropane (a), alongside cyclopropanecarboxaldehyde (b), and cyclopropanemethanol (c). Although cyclopropane lacks a dipole moment and so cannot be detected with radio telescopes, such is not the case with (b) and (c). Aside from astrochemical considerations, cyclopropanecarboxaldehyde, has properties of intrinsic interest. Its gas-phase conformations were shown by Bartell and Guillory to differ from expectations drawn from acetaldehyde, with only two orientations for the former in contrast to the three-fold rotational barrier of the latter.[1]
Here we continue our studies of the IR spectra and properties of small molecules known or suspected to be extraterrestrial. We present new laboratory results on cyclopropanecarboxaldehyde in the solid state, including the first transmission IR spectra of it in an amorphous ice, direct evidence that two conformers are present, a direct measurement of the cis-to-trans ratio of these isomers, and data on the conversion of one isomer into the other. These new results were obtained by recording mid-IR transmission spectra of samples of known thickness and IR path length. A band strength is reported for one IR absorbance of the cis isomer and one for the trans, from which other band strengths can be determined as needed. Our results are compared to older electron diffraction and to computational studies. In addition, we also report the first measurements of several physical properties of amorphous solid forms of cyclopropanecarboxaldehyde and two related molecules. Our cis-to-trans ratio of its conformers trapped from the gas phase is 50:50, close to the 55:45 ratio reported by Bartell and Guillory9, suggesting that the room-temperature distribution of gas-phase isomers was maintained when the cyclopropanecarboxaldehyde vapor condensed into an amorphous solid near 13 K. This appears to be the first such direct comparison between the gas and amorphous-solid phases of this compound. Our results also show that although the trans form is the only isomer present in the polycrystalline solid, such is not the case for amorphous ices.
Mid-IR transmission spectra of amorphous cyclopropanecarboxaldehyde have been presented for the first time. This new data shows that the cis-to-trans abundance ratio of about 1:1 in the gas-phase is maintained when cyclopropanecarboxaldehyde vapor is deposited to form an amorphous solid. The conversion of the cis conformer into the trans structure has been followed and an activation energy measured. An IR band strength for each conformer also has been measured, and the ratio of those band strengths compares favorably to results calculated by others. Density and index of refraction values for amorphous cyclopropane, cyclopropanecarboxaldehyde, and cyclopropanemethanol at 15 K are reported for the first time. The greater stability of the trans isomer over the cis was confirmed in two warmed amorphous ices.
References
[1] Hudson RL, Coleman FM. Solid-State Isomerization and Infrared Band Strengths of Two Conformational Isomers of Cyclopropanecarboxaldehyde, A Candidate Interstellar Molecule. ACS Earth Space Chem. 2019;3(7):1182-1188. doi: 10.1021/acsearthspacechem.9b00058. Epub 2019 May 17. PMID: 31807717; PMCID: PMC6894396.
You may like
Related articles And Qustion
See also
Lastest Price from Cyclopropanecarboxaldehyde manufacturers

US $0.00-0.00/KG2025-04-21
- CAS:
- 1489-69-6
- Min. Order:
- 1KG
- Purity:
- 99%
- Supply Ability:
- 20 mt

US $15.00-10.00/KG2021-07-13
- CAS:
- 1489-69-6
- Min. Order:
- 1KG
- Purity:
- 99%+ HPLC
- Supply Ability:
- Monthly supply of 1 ton