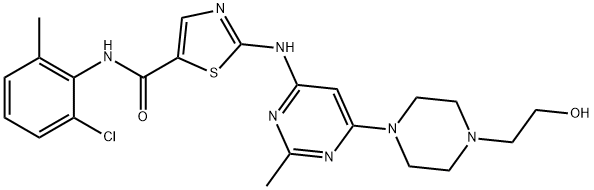
Dasatinib:Brand name,Indication,Side effects and Dosage
In June 2006, the tyrosine kinase inhibitor dasatinib (Sprycel; Bristol-Myers Squibb) was approved by the US FDA for the treatment of adults with chronic myeloid leukaemia and Philadelphia-chromosome-positive acute lymphoblastic leukaemia with resistance or intolerance to previous therapy, including the kinase inhibitor imatinib (Gleevec; Novartis).

Brand name
SPRYCEL
Indication
Dasatinib is a kinase inhibitor indicated for the treatment of:
• newly diagnosed adults with Philadelphia chromosome-positive (Ph+) chronic myeloid leukemia (CML) in chronic phase. The trial is ongoing and further data will be required to determine long-term outcome.
• adults with chronic, accelerated, or myeloid or lymphoid blast phase Ph+ CML with resistance or intolerance to prior therapy including imatinib.
• adults with Philadelphia chromosome-positive acute lymphoblastic leukemia (Ph+ ALL) with resistance or intolerance to prior therapy.
Mechanism of action
Dasatinib is a tyrosine kinase inhibitor with several targets. At nanomolar concentrations, it inhibits BCR-ABL, SRC family (SRC, LCK, YES, FYN), c-KIT, EPHA2, and PDGFRβ. In patients with chronic myeloid leukemia (CML), the tyrosine kinase activity of BCR-ABL is deregulated, leading to the growth, proliferation and survival of cancerous hematopoietic cells. Dasatinib binds to the active and inactive conformation of the ABL kinase domain with a higher affinity than imatinib. In chronic myeloid leukemia (CML) and acute lymphoblastic leukemia (ALL) cell lines overexpressing BCR-ABL, dasatinib inhibits cell growth. Also, dasatinib has in vitro activity against leukemic cell lines that are either sensitive or resistant to imatinib. It has been suggested that dasatinib is able to overcome imatinib resistance caused by BCR-ABL kinase domain mutations because it does not require interaction with some of the residues involved in those mutations.1
Side effects
Most common adverse reactions (≥10%) in patients with newly diagnosed chronic phase CML included myelosuppression, fluid retention, diarrhea, headache, musculoskeletal pain, and rash. Most common adverse reactions (≥20%) in patients with resistance or intolerance to prior imatinib therapy included myelosuppression, fluid retention events, diarrhea, headache, dyspnea, skin rash, fatigue, nausea, and hemorrhage.
Dosage and administration
The recommended starting dosage of dasatinib(SPRYCEL) for chronic phase CML is 100 mg administered orally once daily. The recommended starting dosage of SPRYCEL for accelerated phase CML, myeloid or lymphoid blast phase CML, or Ph+ ALL is 140 mg administered orally once daily. Tablets should not be crushed or cut; they should be swallowed whole. SPRYCEL can be taken with or without a meal, either in the morning or in the evening. In clinical studies, treatment with SPRYCEL was continued until disease progression or until no longer tolerated by the patient. The effect of stopping treatment after the achievement of a complete cytogenetic response (CCyR) has not been investigated.
References
1.Aguilera DG, Tsimberidou AM: Dasatinib in chronic myeloid leukemia: a review. Ther Clin Risk Manag. 2009 Apr;5(2):281-9. Epub 2009 May 4.
You may like
Related articles And Qustion
Lastest Price from Dasatinib manufacturers

US $0.00/g2025-04-21
- CAS:
- 302962-49-8
- Min. Order:
- 1g
- Purity:
- 98%min
- Supply Ability:
- 1000g

US $0.00/KG2025-04-21
- CAS:
- 302962-49-8
- Min. Order:
- 2KG
- Purity:
- USP26, EP VI / GMP DMF
- Supply Ability:
- 20tons