Dasatinib monohydrate:Mechanism,Uses,Side effects and Carcinogenesis
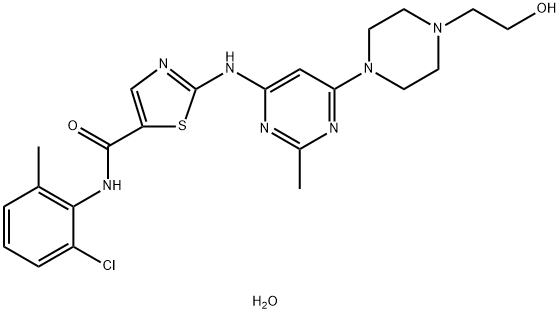
Dasatinib monohydrate is a white to off-white powder, it is a kinase inhibitor.

Mechanism
Dasatinib, at nanomolar concentrations, inhibits the following kinases: BCR-ABL, SRC family (SRC, LCK, YES, FYN), c-KIT, EPHA2, and PDGFRβ. Based on modeling studies, dasatinib is predicted to bind to multiple conformations of the ABL kinase.
Uses
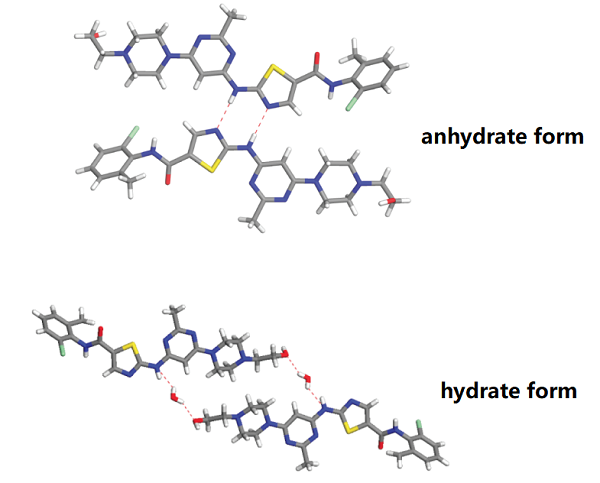
Dasatinib monohydrate indicated for the treatment of chronic myeloid leukemia displays pH-dependent solubility. The aim of reported development program of novel dasatinib anhydrate containing formulation was to demonstrate improved absorption and lower pharmacokinetic variability compared to dasatinib monohydrate.[1]
Side effects
Common adverse effects of Dasatinib monohydrate include low white blood cells, low blood platelets, anemia, swelling, rash, and diarrhea. Severe adverse effects may include bleeding, pulmonary edema, heart failure, and prolonged QT syndrome. Use during pregnancy may result in harm to the fetus. It is a tyrosine-kinase inhibitor and works by blocking a number of tyrosine kinases such as Bcr-Abl and the Src kinase family.
Drug interactions
•CYP3A4 Inhibitors: May increase dasatinib drug levels and should be avoided. If coadministration cannot be avoided, monitor closely and consider reducing SPRYCEL dose.
•CYP3A4 Inducers: May decrease dasatinib drug levels. If coadmini stration cannot be avoided, consider increasing SPRYCEL dose.
•Antacids: May decrease dasatinib drug levels. Avoid simultaneous administration. If needed, administer the antacid at least 2 hours prior to or 2 hours after the dose of SPRYCEL.
•H2 Antagonists/Proton Pump Inhibitors: May decrease dasatinib drug levels. Consider antacids in place of H2 antagonists or proton pump inhibitors.
Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenicity studies were not performed with dasatinib.
Dasatinib was clastogenic when tested in vitro in Chinese hamster ovary cells, with and without metabolic activation. Dasatinib was not mutagenic when tested in an in vitro bacterial cell assay (Ames test) and was not genotoxic in an in vivo rat micronucleus study.
The effects of dasatinib on male and female fertility have not been studied. However, results of repeat-dose toxicity studies in multiple species indicate the potential for dasatinib to impair reproductive function and fertility. Effects evident in male animals included reduced size and secretion of seminal vesicles, and immature prostate, seminal vesicle, and testis. The administration of dasatinib resulted in uterine inflammation and mineralization in monkeys, and cystic ovaries and ovarian hypertrophy in rodents.
References
[1] JIŘÍ HOFMANN. Dasatinib anhydrate containing oral formulation improves variability and bioavailability in humans[J]. Leukemia, 2023, 37 12: 2486-2492. DOI:10.1038/s41375-023-02045-1.
You may like
Related articles And Qustion
Lastest Price from Dasatinib monohydrate manufacturers

US $0.00/kg2025-05-08
- CAS:
- 863127-77-9
- Min. Order:
- 1kg
- Purity:
- 0.99
- Supply Ability:
- 1000kg

US $0.00/KG2025-04-21
- CAS:
- 863127-77-9
- Min. Order:
- 1KG
- Purity:
- 98%min
- Supply Ability:
- 30tons/month