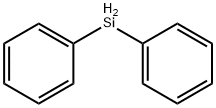
Diphenylsilane as a Organosilicon Compound
Diphenylsilane is an organosilicon compound used as an amide bond coupling reagent & alkene hydrosilylation precursor. Diphenylsilane derivatives can be used for developing blue phosphorescent materials for inkjet printing, which could be used in electronic displays.

The Gas‐Phase Structure and Internal Dynamics of Diphenylsilane
The structure of diphenylsilane could be determined from the rotational spectra of all its singly substituted heavy atom isotopologues. A comparison with its oxygen analogue diphenylether revealed some interesting differences in the internal dynamics as well as in the gas‐phase structure. We report its microwave spectroscopic analysis in the range of 2–8 GHz along with precise gas‐phase structure determination. In addition to the spectroscopic interest, investigating the structural properties of complex organosilicon molecules like diphenylsilane are the basis to its functional behavior, such as the chemical and biological activity. The ability of molecules to adjust their structures according to their chemical environment can be of significant importance for their chemical functioning, often resulting in large‐amplitude motions. For example, in the case of a similar molecule, diphenylether, the molecule undergoes significant structural adjustment to best accommodate the complexing partners like tert‐butyl alcohol. The comparison is then extended to the structural parameters as replacing oxygen with silicon in a molecule leads to an increase of the bond length from C−O to C−Si (due to the larger size of the silicon atom) and can result in structural modification and reactivity. Alongside this, the comprehensive rotational analysis of diphenylsilane and the generated line lists, rotational constants, and centrifugal distortion constants can aid future astronomical searches for diphenylsilane in the interstellar environments. Its detection will unveil an unexplored area of silicon‐substituted aromatic and cyclic molecules.[1]
Diphenylsilane is a near‐prolate asymmetric top molecule with a value of Ray's asymmetry parameter of 𝜅=−0.95 and a calculated electric dipole moment of 0.9 D along the b‐axis. The computed minimum energy structure of diphenylsilane has a C 2 point group symmetry, with the C 2 symmetry axis coinciding with the b‐inertial axis of the molecule. To find the most stable configuration of the molecule, a relaxed dihedral scan of the phenyl ring was performed in steps of 5° at the B3LYP‐D3/aug‐cc‐pVTZ level of theory. The relative energy of the molecule at different values of the dihedral angle is depicted in Figure S1 in the supplementary information. In the most stable configuration the two phenyl rings are arranged in such a way that the dihedral angle described by C1’−C6’−Si−C6 corresponds to approximately 47° to minimize steric hindrance. The comprehensive rotational spectroscopic analysis for this mole‐cule sheds light on its gas‐phase structure and its internal dynamics, which shows that it is rather rigid under the cold conditions of a molecular jet and does not show any splitting in the rotational transitions characteristic of large‐amplitude motion. The rotational spectra of the parent isotopologue of diphenylsilane and all its singly substituted heavy‐atom isotopologues, namely 13C, 29Si, and 30Si, have been assigned with the help of theoretical calculations performed at the B3LYP‐D3/aug‐cc‐pVTZ level of theory.
A good agreement was observed between the theoretical atomic coordinates and the experimentally determined ones. The structural parameters like bond lengths, bond angles, and dihedral angles of diphenylsilane were compared with the structure of its oxygen analogue, diphenylether. This allowed for the identification of changes in the bond angles and bond lengths when the hetero atom is changed from O to Si. This structural comparison was also extended to other silicon containing molecules like rhomboidal silicon tricarbide, trimethylsilyl chloride, phenylsilane, and methyl phenylsilane. As an outlook, the broadband rotational spectroscopy study of diphenylsilane can be potentially extended to high frequency regions, such as the 75–110 GHz range, which overlaps with ALMA Band 3. Such measurements can provide accurate rest frequencies for detecting complex organosilicon molecules like diphenylsilane especially in the regions where other silicon‐containing molecules have been previously detected in high abundances, for example, in the carbon‐rich star IRC+10216. The detection of complex organosilicon molecules will enable us to understand the silicon chemistry taking place in the ISM and its existing chemical complexity.
Non-secosteroidal vitamin D receptor ligands bearing diphenylsilane core
As a platform for investigating the SAR of the hydrophobic structure of non-secosteroidal VDR ligands, we adopted the diphenylsilane framework. Silicon offers several advantages over carbon, including increased hydrophobicity, which is advantageous for promoting hydrophobic interaction between nuclear receptors and their ligands. The Si–C bond is longer than the C–C bond, enabling the construction of sterically crowded quaternary centers. Thus, Si/C-exchange of biologically active compounds has been investigated as an attractive approach to develop novel and distinctive drug candidates. As regards diphenylsilane derivatives, the well-known synthetic method using dichlorosilanes as starting materials can be applied for the preparation of a wide variety of compounds. As hydrogen-bonding moieties of the diphenylsilane-based VDR ligands, we focused on the (S)-4-hydroxypentanoic acid moiety of 4 and the tertiary diethylcarbinol moiety of 10. The (S)-4-hydroxypentanoic acid moiety functions as pharmacophore of potent VDR ligands, and this asymmetric substructure can be readily constructed from a commercially available chiral lactone. The diethylcarbinol moiety is also a promising substructure for binding to VDR without asymmetric complexity. Based on these considerations, we designed diphenylsilane derivatives with diverse hydrophobic core structures.[2]
In this study, we systematically designed and synthesized a series of diphenylsilane derivatives with diverse hydrophobic substituents and investigated their structure–activity relationship (SAR) as VDR agonists. The SAR study revealed that the activity is dependent on the type of substituent and the position of substitution, and the diethyl-di-m-tolylsilane scaffold was identified as the most suitable hydrophobic core structure of this type of VDR ligands. Interestingly, the small structural difference between n-propyl and allyl substituents resulted in a large difference in the activity. Comparison of the co-crystal structures of 14 diphenylsilane compounds, including less potent compounds, bound to the rat VDR ligand-binding domain suggested that the differences in activity are due to a combination of factors, including differences in hydrophilic and hydrophobic interactions, and ligand conformations.
References
[1]Batra, Gayatri, and Melanie Schnell. “Insights Into the Gas-Phase Structure and Internal Dynamics of Diphenylsilane: A Broadband Rotational Spectroscopy Study.” Chemphyschem : a European journal of chemical physics and physical chemistry vol. 26,2 (2025): e202400790. doi:10.1002/cphc.202400790
[2]Mudiyanselage, Hansaka Nirupama Thilakarathne Narasinghe et al. “Structure-activity relationship and crystallographic analyses of non-secosteroidal vitamin D receptor ligands bearing diphenylsilane core as a hydrophobic pharmacophore.” Bioorganic & medicinal chemistry vol. 128 (2025): 118261. doi:10.1016/j.bmc.2025.118261
You may like
See also
Lastest Price from Diphenylsilane manufacturers

US $0.00/KG2025-03-21
- CAS:
- 775-12-2
- Min. Order:
- 1KG
- Purity:
- 98.00%
- Supply Ability:
- 1000KG /month

US $10.00/KG2024-10-11
- CAS:
- 775-12-2
- Min. Order:
- 1KG
- Purity:
- 99.%
- Supply Ability:
- 10 ton