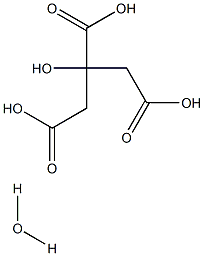
Does citric acid monohydrate dehydrate when heated?
No. Citric acid monohydrate exhibits an inability to desolvate and, upon heating does not dehydrate but undergoes esterification. Nor can it be dehydrated by freeze-drying. The heated sample is not in an anhydrous state; it lacks the ability to melt, and any fluidisation is accompanied by a decomposition process. The increase in molecular weight resulting from the esterification reaction is highly likely to be the reason for the enhanced antioxidant/chelating capacity of the heated sample. What we call citric acid monohydrate and anhydrous citric acid do not exist in pure form (solid state); in reality, they are mixtures composed of varying proportions of citric acid, water, and citric acid oligomers produced by esterification reactions. The esterification reaction appears to proceed readily even under mild heating or at room temperature.
You may like
Related articles And Qustion
See also
Lastest Price from Citric acid monohydrate manufacturers

US $0.00-0.00/KG2025-09-29
- CAS:
- 5949-29-1
- Min. Order:
- 1KG
- Purity:
- 98%min
- Supply Ability:
- 30tons/month

US $1200.00-1100.00/ton2025-08-08
- CAS:
- 5949-29-1
- Min. Order:
- 1ton
- Purity:
- 99%
- Supply Ability:
- 1000T/M