EDTA Magnesium Disodium: Chelate & Material Applications
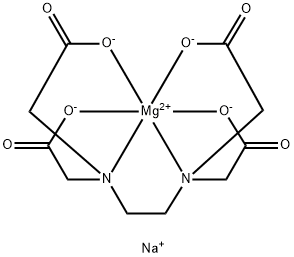
EDTA magnesium disodium is a stable chelate formed by ethylenediaminetetraacetic acid with magnesium and sodium, and often contains water of crystallisation. It appears as a white to pale yellow crystalline powder, is hygroscopic and readily soluble in water, but insoluble in organic solvents such as ethanol. It is resistant to acids, alkalis and high temperatures, with a decomposition temperature exceeding 250°C. As a highly effective chelating agent, it can bind with metal ions such as calcium and iron to form stable, soluble complexes. EDTA magnesium disodium can be used as chelating agent in various fields that binds a wide range of polyvalent cations, including calcium. It also can be used as a nitrogen precursor salt to synthesize nitrogen-doped porous carbon material via direct pyrolysis for supercapacitor applications. EDTA magnesium disodium is well tolerated when running at appropriate doses for over 2-3 hours. Rapid administration will result in severe hypocalcemia leading to hypotension and shock.

Mg-Incorporated Micro-Arc Oxidation Coatings for Orthopedic Implants
Titanium (Ti) and its alloys are widely used as metallic implant materials due to high strength, low density and excellent corrosion resistance, but the materials cannot provide sufficient osseointegration with the surrounding bones in vivo. Hence, implant loosening and failure may occur, and it is sometimes necessary to perform surface treatment to improve the biological properties. In this work, in order to investigate the influences of the electrolyte constituents on the Mg contents and surface properties of anodic coatings, MAO coatings were prepared on Ti-6A1-4V alloys with electrolytes containing 15-g/L H12Phyand different EDTA magnesium disodium (abbreviated as Na2MgY) and potassium hydroxide. The underlying mechanism of the Mg ions incorporation into MAO coatings on titanium alloys was firstly investigated. In addition, the surface characterization and in vitro cytocompatibility of the MAO-treated samples with different Mg contents were evaluated systematically. Widely used medical Ti-6Al-4V alloy samples without annealing or deformation were machined into a cuboidal shape (10 mm× 10 mm × 2 mm or 50 mm × 50 mm × 2 mm). The samples were progressively ground with sandpaper from 80 to 1000 grit, washed with distilled water, dried under hot air and stored in a desiccator prior to MAO treatment. The electrolytes were composed of 15-g/L H12Phyand 5, 10, 15 and 20-g/L EDTA magnesium disodium. In order to study the influences of the solution conductivity on the coating properties, 2, 5, 8 and 11-g/L KOH were separately added into the S-Mg-10 system, a solution with moderate EDTA magnesium disodium concentration.[1]
Larger Na2MgY and KOH concentrations increased the conductivity of the solutions, resulting in a lower breakdown voltage and more spark discharge. Therefore, larger Na2MgY or KOH concentrations enlarge the pore size in anodic coatings and yield a rougher surface morphology. Our study also indicated that EDTA magnesium disodium and KOH influence the surface morphology of MAO coatings in a Na2MgY and KOH concentration-dependent manner. Na2MgY and KOH also affect the chemical composition of anodic coatings. The Na2MgY concentration imposes a larger effect on the Mg and P contents, while KOH shows a larger effect on the Mg content. This influence can be explained according to the MAO characteristics. Similar to magnesium alloys, there are four stages in MAO of titanium alloys—namely, before anodizing, traditional anodizing, MAO treatment and arc anodizing. Ti-6A1-4V alloy samples were subjected to MAO treatment in an environmentally friendlyorganic P-containing and a novel Mg-containing electrolyte and showed good in vitro cytocompatibility based on the viability, adhesion, proliferation and differentiation of the MC3T3-EI pre-osteoblast. Higher EDTA magnesium disodium and KOH concentrations increased the roughness of the MAO coatings, and the best cytocompatibility wasachieved from H12Phy with 10-g/L EDTA magnesium disodium; 5-g/L KOH. H12Phy is a key agent for MAO coating formation, and Mg ions enter into MAO coatings mainly by electromigration. The results suggested that both the surface morphology and Mg contents affected the in vitro cytocompatibility of the MAO coatings on Ti-6A1-4V.
Synthesis of Mg-incorporated ZSM-5 nanosheet via EDTA magnesium disodium
Propylene is a crucial feedstock in chemical industry. It can be used to synthesize some very important chemicals and frequently-used polymers, such as propylene oxide and polypropylene. When it comes to methanol-to-propylene reaction, ZSM-5 is one of the most important catalysts in chemical industry. In our previous work, we had verified the superiority of the ZSM-5 nanosheet compared with the ZSM-5 with irregular morphology and micrometer size, because ZSM-5 nanosheet could provide much shorter diffusion paths. Thus, ZSM-5 nanosheet possessed a long lifetime. In this work, we attempted to use EDTA magnesium disodium (EDTA-MgNa2) to synthesize [Mg]-ZSM-5 nanosheet so that the selectivity of propylene could be improved. Due to the strong chelation between EDTA and Mg2+ ion and the low cost of EDTA-MgNa2, EDTA magnesium disodium was selected to protect Mg2+ ion so that Mg2+ ion could not become Mg(OH)2 precipitation and finally could enter into the framework of ZSM-5. To the best of our knowledge, it was the first time that EDTA-MgNa2 was used as Mg source and [Mg]-ZSM-5 nanosheet was successfully synthesized via one-pot strategy. [Mg]-ZSM-5 nanosheet possessed good physicochemical properties. Especially, [Mg]-ZSM-5 exhibited an excellent catalytic performance in methanol-to-propylene reaction.[2]
Mg-incorporated ZSM-5 nanosheet ([Mg]-Z5-NS) was successfully synthesized via one-pot strategy for the first time and EDTA magnesium disodium was employed as Mg source for the first time because EDTA could protect Mg2+ ions in the course of synthesis. [Mg]-Z5-NS possessed good physicochemical properties. Compared with Z5-NS and Mg/Z5-NS, the selectivity of propylene for [Mg]-Z5-NS-8 wt% increased by 10 % and 4 %, respectively. [Mg]-Z5-NS exhibited an excellent catalytic performance because of its good textural property and distribution of acid sites.
References
[1] RONGFA ZHANG. Novel Mg-Incorporated Micro-Arc Oxidation Coatings for Orthopedic Implants Application.[J]. Materials (Basel, Switzerland), 2021. DOI:10.3390/ma14195710.
[2]Sang, J., Wang, Y., Zhang, X., Su, X., Li, Y., Wen, M., & Ren, G. (2025). One-pot synthesis of Mg-incorporated ZSM-5 nanosheet via EDTA-MgNa₂ as an efficient catalyst for methanol-to-propylene reaction. Chemical Engineering Journal, 520, 165918.
You may like
See also
Lastest Price from EDTA magnesium disodium manufacturers

US $0.00-0.00/G2025-12-29
- CAS:
- 14402-88-1
- Min. Order:
- 1G
- Purity:
- Chelated Magnesium (%) : 5.5% - 6.5%
- Supply Ability:
- 2000

US $0.00-0.00/KG2025-09-28
- CAS:
- 14402-88-1
- Min. Order:
- 1000KG
- Purity:
- 94.5
- Supply Ability:
- 100TONS/MONTH