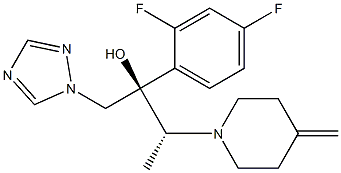
Efinaconazole 10 solution in the treatment of onychomycosis
Onychomycosis is a common, chronic fungal infection of the nail plate or bed that is caused by dermatophytes, nondermatophyte moulds (NDM), or yeasts. While oral antifungals are first-line for moderate-to-severe onychomycosis, topical efinaconazole 10% solution is effective and safe for mild-to-moderate severity onychomycosis. It is FDA-approved for patients aged 6 years and above[1]. This article will introduce its efficacy and safety in older adults[2].
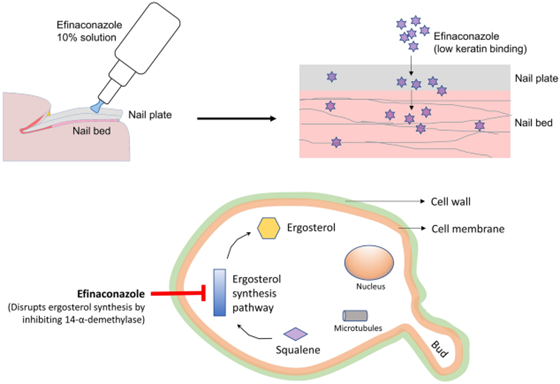
Figure 1. Efinaconazole mechanism of action[3].
Methods
In two multicenter, double-blind, phase 3 studies (NCT01008033; NCT01007708), patients with mild to moderate toenail onychomycosis were randomised (3:1) to once-daily efinaconazole or vehicle for 48 weeks, with a 4-week follow-up. Pooled data for participants aged ≥ 65 years were analysed post hoc (n = 162 efinaconazole, n = 56 vehicle). The primary endpoint was complete cure (0% involvement of target toenail plus mycologic cure [negative KOH and fungal culture]) at week 52. Treatment-emergent adverse events (TEAEs) were assessed throughout.
Efficacy
At week 52, a significantly greater proportion of older adults (aged 65–71 years) achieved complete cure with efinaconazole than vehicle (13.6% vs. 3.6%; p < 0.05). Complete/almost complete cure rate was also significantly greater (≤ 5% involvement and mycologic cure; 19.1% vs. 5.4%; p = 0.01), and over half (59.2%) of participants achieved mycologic cure with efinaconazole versus 12.5% with vehicle (p < 0.001). Treatment-related TEAE rates with efinaconazole were low (6.0%) and similar to the overall study population.
Safety
The proportion of older adults who experienced treatment-emergent AEs (TEAEs) through week 52 was slightly smaller with efinaconazole (67.9% [110/162]) than with vehicle (75.0% [42/56]). The majority of TEAEs were unrelated to the study drug (efinaconazole, 94.0%; vehicle, 98.6%; Table 3), most were mild to moderate in severity, and the discontinuation rate due to AEs was low (< 5%). The most common treatment-related TEAE (reported in ≥ 3% of participants in any treatment group) with efinaconazole was application site dermatitis (4.3%).
Conclusion
Topical efinaconazole 10% solution showed good efficacy in older adults aged 65–71 years with mild to moderate onychomycosis, with a significantly greater percentage of participants achieving complete, complete or almost complete, or mycologic cure versus vehicle at week 52. Additionally, the rates of treatment-related TEAEs and discontinuations due to AEs were low with efinaconazole, demonstrating a good safety profile in this population.
References
[1] Gupta, A. K., Mann, A., Polla Ravi, S., Wang, T., Cooper, E. A. (2024). Efinaconazole 10% solution: a comprehensive review of its use in the treatment of onychomycosis. Expert Opinion on Pharmacotherapy, 25(15), 1983–1998.
[2] Efficacy and Safety of Efinaconazole 10% Topical Solution for Treatment of Onychomycosis in Older Adults: A Post Hoc Analysis of Two Phase 3 Randomised Trials. DOI:10.1111/myc.70069
[3] Gupta, A.K., Talukder, M. Efinaconazole in Onychomycosis. Am J Clin Dermatol 23, 207–218 (2022). DOI:10.1007/s40257-021-00660-1
You may like
Related articles And Qustion
Lastest Price from Efinaconazole manufacturers
US $0.00/kg2025-11-21
- CAS:
- 164650-44-6
- Min. Order:
- 1kg
- Purity:
- 98%
- Supply Ability:
- Customise
US $2.00-5.00/kg2025-06-20
- CAS:
- 164650-44-6
- Min. Order:
- 1kg
- Purity:
- 99%
- Supply Ability:
- 100kg