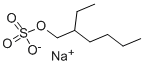
Electrochemical Behavior, Toxicological Profile, and Environmental Treatment of Sodium 2-Ethylhexyl Sulfate
Electrochemical Reduction and Interfacial Adsorption Behavior
The electrochemical reactions of sodium 2-ethylhexyl sulfate (EHS) and its effect on the Zn2+ electroreduction have been investigated at a mercury electrode using cyclic voltammetry. It has been shown that the reduction takes place in two steps. The presence of sodium 2-ethylhexyl sulfate in the solution containing Zn2+ ions moves slightly the potential of zinc reduction towards more negative potentials and causes a slight increase in current density. The differential capacity-potential and differential capacity-time measurements indicate strong adsorption in a wide potential range on the electrode surface. In the potential range −0.46 to −0.86 V vs. saturated calomel electrode and at the concentration lower than the critical micelle concentration (CMC), adsorption for the longer time is hardly reversible. At the concentration higher than the CMC, the formation of hemispherical surface micelles is observed. The theoretical maximum degree of electrode coverage computed with the use of quantum-chemical calculations is equal to 3.53 × 1014 particles cm−2, and it is larger than the value determined experimentally from cyclic voltammograms. In the case of electrochemical reaction, at a current of 0.3 A, during 180 min, the obtained mineralization of sodium 2-ethylhexyl sulfate is only 3%. [1]

Toxicological Effects and Metabolic Fate in Mammals
Sodium 2-ethylhexyl sulfate is marketed under the name of Tergitol anionic surfactant 08, as a 40 % solution in water. Because of some of its applications may result in the toxicity presence of very small amounts in food. Such uses include small amounts in adhesives, in paper and paperboard for packaging dry, fatty or aqueous foods, and the presence of a few tenths of a percent as a penetrant in alkaline solution used to peel fruits and vegetables. Studies reported that the oral LD50 for nonfasted 90-120 g male rats was 10.3 (9.0-11.4) ml/kg of the commercial product; and for nonfasted 250-300 g guinea pigs, 3.8 (3.3-4.2) ml/kg. Fatal doses produced rapid prostration, congestion and hemorrhage of the stomach wall, and bloody urine. When administered to rats for 30 days in the drinking water 0.5% produced no detectable effect, 1% resulted in congested kidneys with dilation of Bowman’s capsule and light cloudy swelling of tubular epithelium, 2% produced additionally albuminuria, and 4% resulted in all these effects as well as reduction of the water voluntarily drunk, but did not retard growth or kill rats. The commercial product produced corneal necrosis in the rabbit eye when applied in volumes greater than 0.005 ml, or when applied as 0.5 ml volumes of concentrations greater than 8 %. Hall (1950) reported on the lung irritation in guinea pigs inhaling aerosols of solutions of the commercial product in water for 8 hr on each of 6 successive days. The minimal histopathologic changes produced by a 1% solution were described. A 0.1% solution produced no changes. The metabolic products from 14C and 35S-labeled samples of 2-ethylhexyl sulfate have been reported by Knaak et al. (1966). Within 4 days of intubation of rats 77 %, of the 14C was collected in urine, 7 % in feces, 7 % in expired air, and none remained in the carcass. Of the labeled material in rat urine, 60 % was unchanged 2-ethylhexyl sulfate, 35-40 % of the sulfur was inorganic, 30 % of the carbon was 2-ethyl-2,3-dihydroxyhexanoic acid, and 6 %, was 2-ethylhexanol and its glucuronide. Identical compounds were found in rabbit urine. [2]
Photocatalytic Degradation and Wastewater Treatment Efficiency
Sodium 2-ethylhexyl sulfate is a kind of surfactant which is widely used in the composition of detergents or care products. Due to the biotoxicity and non-biodegradability, wastewater containing surfactants must be treated before being discharged into the sewage system. Recently the studies found that he efficiency of degradation and mineralization of the anionic surfactant sodium 2-ethylhexyl sulfate depends on physico-chemical parameters such as pH, hydrogen peroxide concentration, catalyst concentration and oxidation time. From the results it was established that the oxidation process of sodium 2-ethylhexyl sulfate in the UV/Fe2+/H2O2 system was carried out with a higher degradation efficiency compared to the UV/TiO2/H2O2 system. The conditions of photo-catalytic oxidation with the Fenton reagent of the anionic surfactant sodium 2-ethylhexyl sulfate were optimized and it was established that it undergoes degradation and mineralization with an efficiency of 95% at [H2O2] = 0.3 mM, [Fe2+] = 0.8 - 1.0 mM, pH = 2.0-2.5 and oxidation time (60 minutes). [3]
References:
[1] Perek-Dlugosz, A., Socha, A., & Rynkowski, J. (2017). Electrochemical reactions of sodium 2-ethylhexyl sulfate salt. Electrocatalysis, 8(3), 270-278.
[2] Smyth Jr, H. F., Carpenter, C. P., Weil, C. S., & King, J. M. (1970). Experimental toxicity of sodium 2-ethylhexyl sulfate. Toxicology and Applied Pharmacology, 17(1), 53-59.
[3] Porubin-Schimb?tor, V., Gonta, M., & Mocanu, L. (2022). Photocatalytic oxidation of the anionic surfactant sodium 2-ethylhexyl sulfate in UV/TiO2/H2O2 and UV/Fe2+/H2O. In Ecological and environmental chemistry (pp. 98-98).
Lastest Price from Sodium 2-ethylhexyl sulfate manufacturers
US $2.00-5.00/kg2025-06-17
- CAS:
- 126-92-1
- Min. Order:
- 1kg
- Purity:
- 99%
- Supply Ability:
- 100kg

US $0.00-0.00/KG2025-04-15
- CAS:
- 126-92-1
- Min. Order:
- 1KG
- Purity:
- 99%
- Supply Ability:
- 500000kg