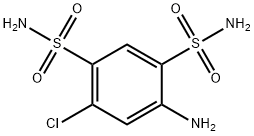
Environmental Pathways, Reactivity, and Synthetic Roles of 4-Amino-6-chlorobenzene-1,3-disulfonamide
Isotopic Synthesis and MS Fragmentation
The synthesis of deuterated thiazides is perfomed by condensation of 4-amino-6-chloro-1,3-benzenedisulfonamide (ABSA) with appropriately labeled aldehydes, while the introduction of 15N into the sulfonamide groups of thiazides was achieved by the synthesis of 4-amino-6-chloro-1,3-benzenedisulfonamide(15N2) from 3-chloroaniline via 4-amino-6-chloro-1,3-benzenedisulfonyl chloride. The most common fragments determined are m/z 269, 205, and 126 for 6-chloro-7-sulfamoyl-3-alkyl-3,4-dihydro-1,2,4-benzothiadiazine-1,1-dioxides and m/z 303, 239, and 160 for 6-trifluoromethyl-7-sulfamoyl-3-alkyl-3,4-dihydro-1,2,4-benzothiadiazine-1,1-dioxides. Individual fragmentation behaviors were found that mainly depended on the C-3-linked side chain. [1]
Thiazide Drug Synthesis Pathways
4-amino-6-chlorobenzene-1,3-disulfonamide can act as an raw material for hydrochlorithiazide and its alkylated analogues. The synthesis of methiazide is described in the following : In a volume of 40 mL of warm ethanol and 6 N aqueous HCl (50:50, v/v) 2.85 g of 4-amino-6-chlorobenzene-1,3-disulfonamide was suspended. A total of 1.5 equiv of acetaldehyde (0.66 g, 840 μL) was added, and the mixture was stirred for 1 h. After storage at 4 °C for 10 h, the precipitate was collected on a medium-porosity frit and washed with bidistilled water until the residue of mineral acid was removed. The resulting methiazide (1.6 g, 52%) was dried in a desiccator over phosphorus pentoxide under reduced pressure. Smaller amounts of synthesized diuretics were extracted from the reaction mixture by dilution with a 5-fold volume of bidistilled water and subsequent solid-phase extraction of the thiazide. To do this, the mixture was transferred onto a PAD-I column (bed height 3 cm, inner diameter 1 cm), washed with distilled water, and the product was eluted with methanol. [1]
Environmental Occurrence and Degradation Pathways of Hydrochlorothiazide and ABSA
Hydrochlorothiazide, HCTZ is frequently used for the treatment of hypertension, congestive heart insufficiency, renal tubular acidosis, prevention of rock formation in kidneys, sometimes even for treatment of hypercalciuria. HCTZ is one the most ubiquitous contaminants in the sewage. In the case of HCTZ hydrolysis only one of degradation product was identified – 4-amino-6-chlorobenzene-1,3-disulfonamide, ABSA have studied photolytic degradation of HCTZ in pure water and sewage treatment plant water with simulated and direct sunlight. Oxidation degradation of HCTZ under harder oxidation conditions (6% H2O2, 80 °C, during 2 h) lead to the formation of 4-Amino-6-chlorobenzene-1,3-disulfonamide. [2]
![Article illustration]()
![Article illustration]() Electronic Structure and Reactivity Analysis
Electronic Structure and Reactivity Analysis
For the 4-Amino-6-chlorobenzene-1,3-disulfonamide molecules ground state geometries have been obtained, higher values of polar surface can be seen. Results indicate much higher reactivity of 4-Amino-6-chlorobenzene-1,3-disulfonamide in water solutions. Analysis of the highest occupied molecular orbital (HOMO) and the lowest unoccupied molecular orbital (LUMO) distribution shows that aromatic rings of 4-Amino-6-chlorobenzene-1,3-disulfonamide have central role. Significantly higher reactivity of HCTZ and 4-Amino-6-chlorobenzene-1,3-disulfonamide can also be addressed to contribution of atomic orbitals of nitrogen and sulfur. [2]
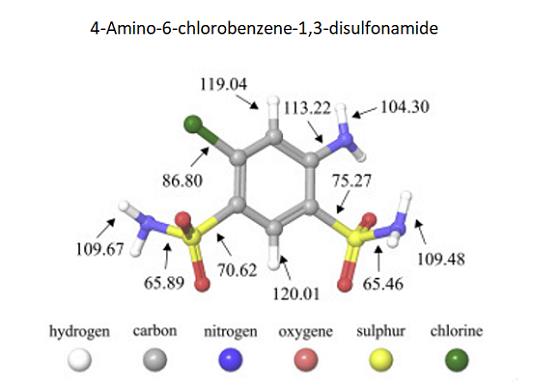
Photocatalytic Degradation and Mechanistic Pathways
Photolytic and photocatalytic degradation of 4-Amino-6-chlorobenzene-1,3-disulfonamide in the presence of TiO2 under different sources of irradiation was investigated. Fundamental reactive properties of HCTZ and 4-Amino-6-chlorobenzene-1,3-disulfonamide in water solution have been obtained by DFT and MD simulations. Various global and local reactivity properties such as dipole moment, ALIE, solvation free energies, (H-)BDE, RDFs, have been considered, including the ring opening investigation by transition state search. Results have indicate that higher reactivity of 4-Amino-6-chlorobenzene-1,3-disulfonamide, and especially of HCTZ in water solution comparing to beta-blockers, is consequence of the significant charge separation within molecule and strong interactions of hydrogen atoms, connected to nitrogen atoms, with water molecules. Under UVA irradiation already after 25 min 4-Amino-6-chlorobenzene-1,3-disulfonamide was completely removed from water solution, while under SSI after more than 120 min 4-Amino-6-chlorobenzene-1,3-disulfonamide was present in solution at an optimal mass concentration of catalyst of 2.0 mg/mL and 0.05 mmoL/L start concentration of 4-Amino-6-chlorobenzene-1,3-disulfonamide. After 240 min under SSI 65% of mineralization was achieved. In the investigated interval of concentrations of 4-Amino-6-chlorobenzene-1,3-disulfonamide (0.01–0.1 mmoL/L), the efficiency of photocatalytic degradation decreases with the increase of substrate concentration under SSI, but under UVA irradiation optimal initial concentration of 4-Amino-6-chlorobenzene-1,3-disulfonamide has been achieved for 0.025 mmoL/L. Four intermediates were detected applying LC‐ESI‐MS/MS technique. [2]
References:
[1] Thevis, M., Schmickler, H., & Sch?nzer, W. (2002). Mass spectrometric behavior of thiazide-based diuretics after electrospray ionization and collision-induced dissociation. Analytical chemistry, 74(15), 3802-3808.
[2] Armakovi?, S. J., Armakovi?, S., ?etojevi?-Simin, D. D., ?ibul, F., & Abramovi?, B. F. (2018). Photocatalytic degradation of 4-amino-6-chlorobenzene-1, 3-disulfonamide stable hydrolysis product of hydrochlorothiazide: Detection of intermediates and their toxicity. Environmental Pollution, 233, 916-924.
See also
Lastest Price from 4-Amino-6-chlorobenzene-1,3-disulfonamide manufacturers

US $0.00-0.00/Kg/Drum2025-04-21
- CAS:
- 121-30-2
- Min. Order:
- 1KG
- Purity:
- 98%min
- Supply Ability:
- 500kg
US $0.00-0.00/mg2025-04-18
- CAS:
- 121-30-2
- Min. Order:
- 10mg
- Purity:
- 98
- Supply Ability:
- 100000