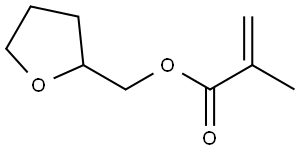
Free-Radical Copolymerization and Chemical Applications of Tetrahydrofurfuryl methacrylate
Tetrahydrofurfuryl methacrylate is a colorless transparent liquid under ambient temperature and pressure, possessing a distinct pungent odor and high chemical reactivity. Primarily utilized as a fundamental building block in organic chemical processes, tetrahydrofurfuryl methacrylate is widely employed in the industrial production of ink polymers, two‑component acrylic adhesives, auxiliary cross‑linking agents for rubber and plastics, rubber modifiers, as well as photopolymers and photoresists.

Figure1: Picture of Tetrahydrofurfuryl methacrylate
Chemical Properties
Tetrahydrofurfuryl methacrylate is a monomer containing an acrylate group. It can undergo free‑radical copolymerization with other acrylic monomers to form polymeric materials. Tetrahydrofurfuryl methacrylate also exhibits good chemical stability and can withstand certain acidic and alkaline conditions. With its high boiling point, low viscosity, and cyclic alkyl group, this compound is mainly used in anaerobic adhesives, wire and cable coatings, screen‑printing inks, rubber modifiers, emulsion polymers, plastic products, artificial nails, and related applications.
Free-Radical Copolymerization
The free‑radical copolymerization of hydroxyethyl methacrylate (HEMA) and tetrahydrofurfuryl methacrylate (THFMA) was investigated at 50 °C. The compositions of polymers obtained at low conversions were determined by 13C‑NMR, and their glass‑transition temperatures were measured using DSC. The copolymerization behavior was best described by a terminal model with reactivity ratios of rH = 1.79 and rT = 0.76. Triad‑fraction sequence distributions were calculated based on this terminal model and the derived reactivity ratios. The glass‑transition data were fitted to the Gordon–Taylor equation, yielding an optimal Gordon–Taylor constant of kH = 1.42 ± 0.2, which indicates non‑ideal mixing of the two monomer components in the copolymers. [1]
Chemical Applications
Tetrahydrofurfuryl methacrylate is widely used in the coatings and inks industry as a thickener, solvent, and modifier to enhance fluidity, adhesion, and abrasion resistance. It can copolymerize with other acrylic monomers to form acrylic resins, which are commonly employed in UV‑curable coatings, adhesives, and resin modifications, offering excellent chemical resistance and physical properties. Additionally, tetrahydrofurfuryl methacrylate finds application in organic synthesis as an intermediate for the preparation of various compounds such as polymers, pharmaceuticals, and dyes. In the pharmaceutical field, due to its good biocompatibility, tetrahydrofurfuryl methacrylate is also utilized in certain biomedical applications.
Phase behavior
A comprehensive set of high-pressure phase behavior data for the binary carbon dioxide/tetrahydrofurfuryl methacrylate (CO₂/THFMA) system is presented, covering a temperature range of 313.2 to 393.2 K and pressures up to 22.07 MPa, where the system exhibits classical Type-I phase behavior characterized by a continuous mixture-critical curve. Furthermore, the solubility of poly(tetrahydrofurfuryl methacrylate) [P(THFMA)] in supercritical carbon dioxide was investigated in the presence of either THFMA or dimethyl ether (DME) as a cosolvent, with measurements extending to severe conditions of 490.5 K and 289.83 MPa. It was observed that the cloud-point pressure for the P(THFMA) + CO₂ system markedly decreases upon the addition of either THFMA or DME as a cosolvent, whereas P(THFMA) proved insoluble in pure CO₂ even under extreme conditions of 498 K and 300.0 MPa. All experimental results, encompassing the vapor-liquid equilibrium (VLE) data for the CO₂/THFMA binary system and the cloud-point phase behavior for the P(THFMA)/CO₂/cosolvent ternary systems, were successfully correlated using the Perturbed-Chain Statistical Associating Fluid Theory (PC-SAFT) equation of state. Subsequent detailed calculations performed with the PC-SAFT model revealed that the underlying mechanism for the observed upper critical solution temperature (UCST)-type phase behavior is predominantly governed by the interactions between like molecules—specifically, those between THFMA and THFMA and between CO₂ and CO₂—rather than by the cross-interactions between the different species, namely between THFMA and CO₂. In addition, the critical-point (CP), bubble-point (BP), and dew-point (DP) pressures for the CO2/THFMA system were measured at temperatures in the range of(313.2–393.2)Kandatpressuresupto22.07MPa.[2]
References
[1] Ghi, P. Y.; Hill, D. J. T.; Whittaker, A. K. A study of the copolymerization of hydroxyethyl methacrylate and tetrahydrofurfuryl methacrylate. J. Polym. Sci. Part A: Polym. Chem. 1999, 37, 3730–3737.
[2] Cho S H, Lee B S, Byun H S. Phase behavior of tetrahydrofurfuryl methacrylate and poly (tetrahydrofurfuryl methacrylate) in supercritical carbon dioxide[J]. Journal of CO2 Utilization, 2018, 25: 39-45.
You may like
Lastest Price from Tetrahydrofurfuryl methacrylate manufacturers

US $79.00-38.00/kg2025-04-21
- CAS:
- 2455-24-5
- Min. Order:
- 1kg
- Purity:
- 99%
- Supply Ability:
- 20ton

US $0.00/kg2025-04-15
- CAS:
- 2455-24-5
- Min. Order:
- 20kg
- Purity:
- 99.0%
- Supply Ability:
- 10 tons