H-Lys(Boc)-OH: Protected Lysine for Peptide & Antibiotic Synthesis
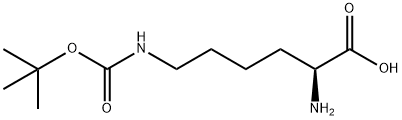
H-Lys(Boc)-OH is a key amino acid derivative for the selective protection of the side-chain amino group of L-lysine. Within the molecule, only the ε-amino group of lysine is protected by Boc (tert-butoxycarbonyl), while the α-amino group and carboxyl group remain free to directly participate in peptide bond condensation. This makes it a core building block for controlling lysine sites in peptide synthesis. In both solid-phase and liquid-phase peptide synthesis, it prevents side-chain amino group side reactions, enabling precise sequence control. H-Lys(Boc)-OH is widely employed in the preparation of anti-tumour, anti-infective, peptide drug intermediates, and bioactive peptides. The Boc protecting group can be removed under mild acidic conditions, offering high compatibility, straightforward handling, and minimal impact on the peptide backbone. H-Lys(Boc)-OH finds application in chemical biology, protein modification, and the construction of antibody-drug conjugate linkers. As an indispensable protected amino acid in drug discovery and organic synthesis, it typically requires storage at low temperatures, in the dark, and under sealed conditions.

H-Lys(Boc)-OH used in chemical synthesis
Angiogenesis is described as the formation of new blood vessels from pre-existing vessels. It is a vital physiological process for growth, development, and wound repair by which various materials, oxygen and nutrients are delivered to tissues and cellular wastes are removed. In cancer, abnormal angiogenesis is related to cell growth, tumorigenesis, and metastases. In previous research efforts by our group, the cyclic pentapeptide c(RGDyK), which is a potent integrin αvβ3 ligand, was employed for the targeted delivery of various anticancer drugs and bioactive molecules such as gemcitabine, the triterpenoids cucurbitacins, and platinum complexes. In this work, the synthesis of JH-VII-139-1 peptide conjugates with c(RGDyK) and the properties of the new hybrid compounds against SRPK1, cancer cell growth and angiogenesis are reported. During the above complex synthetic process, H-Lys(Boc)-OH is added as an intermediate: To a stirred solution (11 mg, 15.3 μmol) and H-Lys(Boc)-OH (3.8 mg, 15.3 μmol) in DMF (0.8 mL) was added triethylamine (8.5 μL, 61 μmol) at ambient temperature, under Ar. The mixture was stirred at room temperature for 24 h. Subsequently, the solvent was evaporated, and the product was purified with flash column chromatography (gradient elution DCM–MeOH 10:1, 10:2, 10:3) to yield (S)-14-(4-((tert-butoxycarbonyl)amino)butyl)-1-(3-(3-cyano-9-ethyl-6,6-dimethyl-11-oxo-6,11-dihydro-5H-benzo[b]carbazol-8-yl)-1H-pyrazol-1-yl)-1,12-dioxo-5,8-dioxa-2,11,13-triazapentadecan-15-oic acid (8 mg, 63%) as white solid. Lysine was a good candidate, as it could extend the triethylene glycol chain and bear both a carboxylic and an amino group. This approach that eventually led to the synthesis of conjugate geo107. The synthesis starts with a nucleophilic attack by H-Lys(Boc)-OH to the highly reactive 4-nitrophenyl carbamate that produces Boc-protected product in 63% yield. [1]
H-Lys(Boc)-OH for Production of Modified Lantibiotics
Lantibiotics represent a class of peptide antibiotics that shows promising antimicrobial activity against Gram-positive pathogens, e.g., Staphylococcus aureus or Clostridium difficile . The best studied example is nisin, synthesized by the Gram-positive bacterium Lactococcus lactis. It belongs to the class of RiPPs and is translated as a precursor comprising a 23 amino acid leader peptide fused to a 34 amino acid core peptide. To specifically introduce the ncAA at the desired position in the protein of interest, we focused on implementing the SCS method (Wang et al., 2001) in L. lactis. This technology allows reprogramming the amber stop codon TAG to a sense codon for the ncAA. Genome analysis revealed TAG to be the least frequently used stop codon in L. lactis (Gupta et al., 2004). In this study, a double tracked approach toward ncAA-modified nisin was followed: First, E. coli was equipped with both the SCS and nisin biosynthesis machineries for recombinant production of nisin modified with H-Lys(Boc)-OH (BocK) as chosen ncAA. Second, the genetic code of L. lactis, the Gram-positive native nisin production host, was expanded by introducing the PylRS–tRNAPyl pair to enable SCS. Following site-specific incorporation of H-Lys(Boc)-OH into different locations of nisin in vivo, the effects on the antimicrobial activity of nisin were analyzed.[2]
Two internal negative controls were performed. Supernatant of cells expressing only PylRS–tRNAPyl either in the presence and absence of BocK never had any antimicrobial effect. Consequently, antimicrobial activity was caused by the novel nisin variants and not by the orthogonal tRNA synthetase–tRNA pair or the supplemented ncAA. In this study, we established genetic code expansion of L. lactis by functional expression of an OTS based on PylRS–tRNAPyl. The successful combination of SCS and PTM enzymes led to the production of bioactive H-Lys(Boc)-OH -modified nisin variants. To identify the ideal host for genetic code expansion and post-translationally modified lantibiotic production, we tested two expression hosts: E. coli and L. lactis, because each of them has distinct advantages. The incorporation of non-canonical amino acids (ncAAs) into ribosomally synthesized and post-translationally modified peptides, e.g., nisin from the Gram-positive bacterium Lactococcus lactis, bears great potential to expand the chemical space of various antimicrobials. The H-Lys(Boc)-OH (BocK) was chosen for incorporation into nisin using the archaeal pyrrolysyl-tRNA synthetase–tRNAPyl pair to establish orthogonal translation in L. lactis for read-through of in-frame amber stop codons. In parallel, recombinant nisin production and orthogonal translation were combined in Escherichia coli cells. Both organisms synthesized bioactive nisin(H-Lys(Boc)-OH) variants. Screening of a nisin amber codon library revealed suitable sites for ncAA incorporation and two variants displayed high antimicrobial activity. Orthogonal translation in E. coli and L. lactis presents a promising tool to create new-to-nature nisin derivatives.
References
[1]Leonidis G. et al. Synthesis and Anti-Angiogenic Activity of Novel c(RGDyK) Peptide-Based JH-VII-139-1 Conjugates. Pharmaceutics 2023, 15(2):381.
[2]Bartholomae M. et al. Expanding the Genetic Code of Lactococcus lactis and Escherichia coli to Incorporate Non-canonical Amino Acids for Production of Modified Lantibiotics. Frontiers in Microbiology 2018.
Lastest Price from H-Lys(Boc)-OH manufacturers

US $0.00-0.00/kg2025-04-21
- CAS:
- 2418-95-3
- Min. Order:
- 1kg
- Purity:
- 98%
- Supply Ability:
- 1T+

US $0.00/kg2023-07-27
- CAS:
- 2418-95-3
- Min. Order:
- 1kg
- Purity:
- 99%
- Supply Ability:
- 2T/month