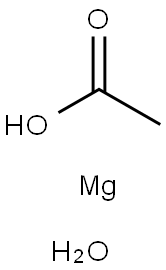
How does magnesium acetate tetrahydrate form anhydrous glass?
The thermal dehydration of magnesium acetate tetrahydrate is known as a process which produces noncrystal line anhydrate. The thermal dehydration of magnesium acetate tetrahydrate begins at the surface of the reaction particles; the mechanism involves the melting and/or dissolution of the ionic material in the water produced, thereby forming a fluid phase. Research has shown that the formation of this intermediate fluid phase is a characteristic phenomenon leading to the formation of anhydrous glass. Upon further heating, the newly prepared anhydrous glass exhibits a glass transition at approximately 470 K, with an activation energy Ea ≈ 550–560 kJ mol–1, followed by crystallisation via diffusion-controlled three-dimensional growth of crystal nuclei. The activation energy for this crystallisation process is approximately 280 kJ mol–1, with an enthalpy change of ΔH = –13.3 kJ mol–1, ultimately resulting in the formation of small, rounded crystalline anhydrous particles.
You may like
Related articles And Qustion
See also
Lastest Price from Magnesium acetate tetrahydrate manufacturers

US $0.00-0.00/kg2026-03-11
- CAS:
- 16674-78-5
- Min. Order:
- 100kg
- Purity:
- 99%
- Supply Ability:
- 20 tons per day

US $1200.00-1100.00/ton2025-08-11
- CAS:
- 16674-78-5
- Min. Order:
- 1ton
- Purity:
- 99%
- Supply Ability:
- 1000T/M