Is iodine an oxidising agent?
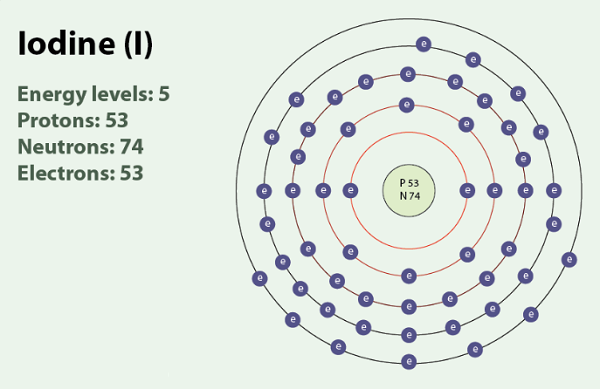
Yes. Iodine is a chemical element with atomic number 53 and a mass number ranging from 108 to 144. Only iodine-127 (with an atomic mass of 126.9) is stable; all other isotopes of iodine are radioactive. Iodine (I) lacks one electron to achieve a complete octet, and is therefore a strong oxidising agent. The element iodine forms diatomic molecules (I2), in which two iodine atoms share a pair of electrons to achieve a stable octet structure. Iodine has oxidation states ranging from -1 to +7. The iodide ion (I⁻) is the strongest reducing agent among the stable halogens and is the most readily oxidised back to the diatomic iodine molecule (I2). Iodine in the form of I2 is a solid with a melting point of 113.7 °C; it sublimates readily upon slight heating.
Related articles And Qustion
See also
Lastest Price from iodine manufacturers

US $360.00-99.00/kg2025-04-21
- CAS:
- 12190-71-5
- Min. Order:
- 1kg
- Purity:
- 99% purity,WhatsApp+86 18102676775
- Supply Ability:
- 20 tons

US $23.00-1.00/kg2025-03-07
- CAS:
- 12190-71-5
- Min. Order:
- 1kg
- Purity:
- 99%
- Supply Ability:
- 300tons