Mechanism of Action and Pharmacokinetics of Oxolinic acid
Oxolinic acid (5-ethyl-5,8-dihydro-8-oxo-1,3-dioxolo[4,5-g]quinoline-7-carboxylic acid) is a synthetic antimicrobial agent approved by the Food and Drug Administration for the treatment of bacterial urinary tract infections in adults that are established and not associated with bacteremia, and Oxolinic acid's chemical structure, mechanism of action, spectrum of activity, and potential for toxicity are similar to those of nalidixic acid.

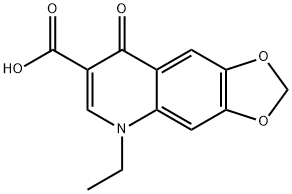
Figure1: Picture of Oxolinic acid
Basic Introduction
Oxolinic acid is a second-generation quinolone bactericide. At room temperature, it appears as white to yellowish-white columnar crystals or crystalline powder, odorless and tasteless. It exhibits strong antibacterial efficacy against Gram-negative bacteria and some Gram-positive bacteria, and shows no cross-resistance with antibiotics. It has the advantages of low dosage and good bacteriostatic effects. Oxolinic acid is primarily used to treat bacterial diseases in fish and shrimp, and is considered by aquaculture practitioners to be an ideal drug for treating diseases in aquatic animals. It has considerable antibacterial activity against fish pathogens such as Vibrio anguillarum and Aeromonas hydrophila.[1]
Mechanism of Action
Given the structural similarities between oxolinic acid and nalidixic acid, their mechanism of action appears to be identical, involving interference with deoxyribonucleic acid (DNA) synthesis. Oxolinic acid, however, demonstrates a tenfold greater capacity to inhibit DNA replication, and this enhanced potency is reflected in its increased in vitro activity against Enterobacteriaceae.
Spectrum of Activity
Oxolinic acid demonstrates in vitro activity against most Gram-negative aerobic urinary tract pathogens, with the majority of Escherichia coli, Klebsiella sp., Enterobacter sp., and Proteus sp. being susceptible to oxolinic acid. Oxolinic acid also exhibits in vitro activity against Salmonella sp., Shigella sp., Neisseria meningitidis, Neisseria gonorrhoeae, and Staphylococcus aureus. However, Pseudomonas aeruginosa and Acinetobacter calcoaceticus (both var. lwoffi and var. anitratus) are uniformly resistant to oxolinic acid, and it has no activity against other Gram-positive aerobic cocci. Oxolinic acid demonstrates susceptibility against Salmonella sp., Shigella sp., and Neisseria meningitidis and Neisseria gonorrhoeae. Additionally, oxolinic acid possesses in vitro activity against Staphylococcus aureus, although it has no activity against other Gram-positive aerobic cocci.
Pharmacokinetics
After oral administration, oxolinic acid is absorbed rapidly from the gastrointestinal tract. Peak serum concentrations of the biologically active, unconjugated drug are attained in two to four hours and range from 1.8 to 3.6 μg/ml. Oxolinic acid reaches steady-state serum concentrations in three days, after which no further drug accumulation occurs, with lower serum levels of unconjugated drug observed during the first three days of dosing, suggesting a slow distribution into tissue pools; however, no data on the details of oxolinic acid distribution in humans have been published. Protein binding of oxolinic acid is about 77 to 81%. The completeness of oxolinic acid absorption is not clearly established, as daily urinary recovery of oxolinic acid and its metabolites accounts for only 43 to 49% of administered drug, while fecal excretion accounts for 16 to 20%, which could represent either incomplete absorption or biliary excretion. After repeated dosing with oxolinic acid, a two-phase elimination pattern has been observed in humans, with an initial rapid distribution phase having a half-life of about 1.5 hours, followed by a slow elimination phase with a half-life of about 15 hours. Urinary concentrations of unconjugated drug range from 15 to 155 μg/ml in patients with normal renal function, and urinary excretion is apparently independent of pH. Unconjugated oxolinic acid accounts for only 1.4 to 21% of drug excreted in the urine, while glucuronide and non-glucuronide metabolites account for greater than 80%. Renal insufficiency results in increased serum concentrations of unconjugated oxolinic acid (5.5 to 6.1 μg/ml) and only minimally decreased urinary levels (35 to 76 μg/ml); therapeutic urinary concentrations are achieved even in the presence of severe renal impairment, and excessive drug accumulation has not been observed in azotemic patients. There are no reports of clinically important drug interactions or alterations of laboratory test values. [1]
Adverse Reactions
A reliable estimate of the nature and frequency of adverse reactions to oxolinic acid is unavailable because the drug has been the subject of only a few published reports, with reports submitted to the manufacturer constituting essentially the only toxicity data—a method of surveillance that is notoriously unreliable. Published studies suggest that adverse reactions are experienced by 10 to 45% of oxolinic acid recipients, the most common being central nervous system and gastrointestinal disturbances, which are usually mild and resolve with discontinuation of treatment. Oxolinic acid necessitates cessation of therapy for approximately 5% of patients due to adverse reactions. Central nervous system toxicity has included insomnia (the most frequent), restlessness, dizziness, headache, drowsiness, and visual disturbances. In contrast to nalidixic acid, oxolinic acid has not been implicated as causing intracranial hypertension, and an association with seizures has not been clearly established; however, oxolinic acid has a stimulatory effect on the central nervous system and should not be prescribed for patients with known seizure disorders. Gastrointestinal reactions include nausea (the most frequent), vomiting, abdominal discomfort, and diarrhea, which can be minimized by concurrent administration of food or antacids. [1]
Therapeutic Use
A very limited number of published studies provide information on the value of oxolinic acid in bacterial urinary tract infections, and unfortunately, few of these investigations are adequate methodologically. The data derived from reasonably well-designed investigations suggest that oxolinic acid is effective for the therapy of acute and recurrent urinary tract infections in adults caused by Escherichia coli and Proteus mirabilis; that oxolinic acid is no more effective or better tolerated than other drug treatments such as trimethoprim-sulfamethoxazole, ampicillin, and nalidixic acid; and that the therapeutic efficacy of oxolinic acid is not dose related. Oxolinic acid has a marked propensity to evoke resistant mutants when the daily dose is less than 2 g, but when prescribed in the full therapeutic dose of 2 g/day, it does not evoke a greater incidence of resistant mutants than nalidixic acid. Oxolinic acid is an effective prophylactic agent for adult women with symptomatic recurrent urinary tract infections, and is also an effective therapeutic agent for children with acute or recurrent bacterial urinary tract infections; however, oxolinic acid is not currently approved by the Food and Drug Administration for these indications. Although oxolinic acid has in vitro activity against chloramphenicol-resistant strains of Salmonella typhi, it is not useful in the treatment of infections caused by this organism, and while oxolinic acid has shown clinical and bacteriological efficacy when administered to adult volunteers who developed shigellosis, this is not an approved indication either.
Therapeutic Indications
Oxolinic acid serves as an alternative form of therapy for penicillin-sensitive, cephalosporin-sensitive, or sulfonamide-sensitive adults with recurrent urinary tract infections caused by susceptible Escherichia coli or Proteus mirabilis that are not complicated by bacteremia. Oxolinic acid should not be prescribed for patients with acute symptomatic urinary tract infections, patients experiencing bacteremia, pregnant women, or children.
Synthesize neutral mononuclear zinc complexes
Oxolinic acid, a quinolone antibacterial drug, was used as a ligand to synthesize and characterize neutral mononuclear zinc complexes in the absence or presence of the nitrogen-containing heterocyclic ligands 2,2′-bipyridine or 1,10-phenanthroline. Experimental data indicate that oxolinic acid acts in its deprotonated form as a bidentate ligand, coordinating to the metal ion through the ketone oxygen and one carboxylate oxygen atom. Oxolinic acid was further employed in the determination of the crystal structures of (chloro)(oxolinato)(2,2′-bipyridine)zinc(II) complex 2 and bis(oxolinato)(1,10-phenanthroline)zinc(II) complex 3 via X-ray crystallography, and the biological activity of the complexes was evaluated by examining their binding ability to calf-thymus DNA through UV and fluorescence spectroscopy. [2]
Reference
[1] Gleckman R, Alvarez S, Joubert D W, et al. Drug therapy reviews: oxolinic acid[J]. American Journal of Hospital Pharmacy, 1979, 36(8): 1077-1079.
[2] Tarushi A, Psomas G, Raptopoulou C P, et al. Zinc complexes of the antibacterial drug oxolinic acid: structure and DNA-binding properties[J]. Journal of inorganic biochemistry, 2009, 103(6): 898-905.
You may like
Related articles And Qustion
Lastest Price from Oxolinic acid manufacturers

US $51.00/box2025-09-28
- CAS:
- 14698-29-4
- Min. Order:
- 1box
- Purity:
- 99%
- Supply Ability:
- 20tons

US $0.00-0.00/Kg/Drum2025-04-21
- CAS:
- 14698-29-4
- Min. Order:
- 1KG
- Purity:
- 98%-102%
- Supply Ability:
- 20 tons