Mechanism of Action of Lenalidomide in the Treatment of Multiple Mieloma
Lenalidomide is a second‑generation new immuno modulatory medication used to treat multiple myeloma (MM). Its mechanism of action involves affecting the expression of vascular endothelial growth factor, interleukin‑6, cytochrome c, caspase‑8, as well as other factors including immunological modulation and the direct killing of cells, among others, rendering it a fundamental medication, useful for the treat ment of MM.
Mechanism of Action
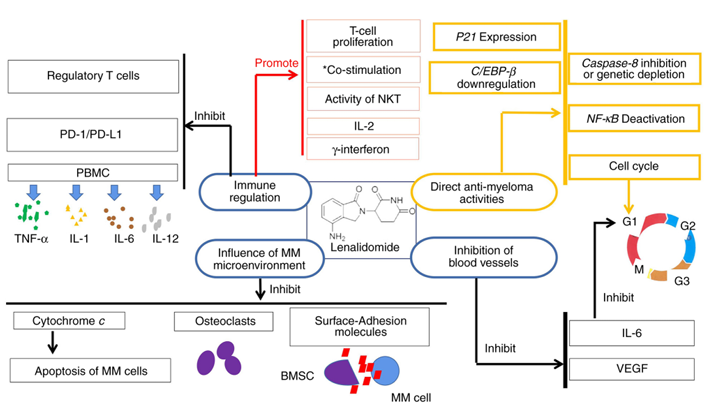
Lenalidomide, a second‑generation imine medication, is relatively less toxic and with a higher potency when compared with thalidomide. Lenalidomide has been demonstrated to exhibit an array of effects and mechanisms of action that can contribute to its antitumor properties (Fig. 1).
Non‑immune regulation
Vascular endothelial growth factor (VEGF) is inhibited by lenalidomide, which makes it chal lenging for tumor cells to form blood vessels. It blocks VEGF and can prevent the production of interleukin‑6 (IL‑6). As per a previous study, IL‑6 is a cytokine with a wide range of inflammatory and immune regulatory properties. In addi tion, IL‑6 can promote the progression of MM. Lenalidomide has also been linked to the growth arrest of myeloma cells in the G1 phase, and this direct cytotoxicity is associ ated with a decrease in IL‑6 production. However, the precise mechanism underlying this effect is unknown.
A well‑known mitochondrial protein, cytochrome c, can maintain life by transporting electrons to the respiratory chain and allowing continued ATP production. Cell survival and apoptosis significantly depend on cytochrome c. By influencing the release of cytochrome c, lenalidomide can impact the apoptosis of MM cells. In addition, by altering caspase‑8, lenalidomide can also affect the apoptosis of MM cells.
Lenalidomide can directly induce MM cell apoptosis and cell cycle arrest. Previous studies have demonstrated several downstream changes after lenalidomide treatment, which may be associated with the direct anti‑myeloma activities of the drug, in addition to the previously mentioned mechanism. These changes include the upregulation of P21 expression, nuclear factor‑κB (NF‑κB) deactivation, CCAAT/enhancer binding protein‑β (C/EBP‑β) downregula tion, and caspase‑8 inhibition or genetic depletion.
By suppressing the production of surface‑adhesion molecules on both MM cells and bone marrow stromal cells (BMSCs), lenalidomide prevents contact between them . Lenalidomide can prevent MM‑related bone damage by either directly preventing osteoporosis development or by indirectly decreasing the tumor load. The effect of lenalidomide on osteoclasts can slow the development of MM, as osteoclasts have been demonstrated to increase MM growth and medica tion resistance.
Immune regulation
In contrast to other anti‑MM medications, lenalidomide possesses immunoregulatory effects. First, it can improve the co‑stimulation of CD4+ and CD8+ T cells. When compared to the first‑generation IMiD thalidomide, lenalidomide increases T‑cell proliferation and the production of IL‑2 and γ‑interferon. Lenalidomide can also inhibit regulatory T cells, which are a subset of immunosuppressive T cells, that are important for self‑tolerance and the reaction of the the immune system to tumor cells. With the natural killer (NK) cell‑surface markers, lenalidomide increases the activity of NK and T lymphocytes (NKT). In patients with MM who have received suitable treatment, NK cell proliferation is promoted, an important pharmacological effect of lenalidomide. It has also been demonstrated that the ability of human peripheral blood mononuclear cells (PBMC) to produce the pro‑inflammatory cytokines tumor necrosis factor‑α (TNF‑α), IL‑1, IL‑6, and IL‑12 is inhibited by the effect of lenalidomide. Lenalidomide reduces the immune checkpoint inhibitor programmed death‑1 (PD‑1) expression on both T and NK cells in patients with MM; it also reduces the expression of PD‑1 and programmed death ligand‑1 (PD‑L1) on MM cells.
References:
[1] CHAO-WEI ZHANG; Xue L G; Ya Nan Wang. Lenalidomide use in multiple myeloma (Review).[J]. Molecular and clinical oncology, 2023, 20 1: 7. DOI:10.3892/mco.2023.2705.
You may like
Related articles And Qustion
See also
Lastest Price from Lenalidomide manufacturers

US $0.00/KG2025-09-26
- CAS:
- 191732-72-6
- Min. Order:
- 1KG
- Purity:
- ≥99.9%(HPLC)
- Supply Ability:
- 10000KGS

US $5.00-0.50/KG2025-05-08
- CAS:
- 191732-72-6
- Min. Order:
- 1KG
- Purity:
- 99% hplc
- Supply Ability:
- 500TONS