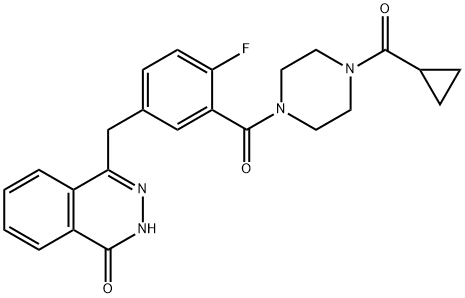
Mechanism of action of olaparib in the treatment of ovarian cancer
Olaparib is used to treat BReast CAncer susceptibility protein (BRCA)-associated, platinum-sensitive ovarian cancer. Olaparib inhibits poly(ADP-ribose) polymerase, thereby blocking the repair of single-strand DNA breaks. This results in synthetic lethality in BRCA-associated cancer cells, which have a dysfunction of another DNA repair pathway– homologous recombination.
Indication
Olaparib is indicated in adult patients with high-grade serous epithelial ovarian, fallopian tube and primary peritoneal
cancer. The EuropeanMedicinesAgencyhasapprovedolaparibformaintenancetreatmentoftumoursthatarebothBRCA
mutated and platinum sensitive (currently in response to last platinum therapy and ≥6 month duration of progression
free survival after penultimate platinum therapy).
Mechanism
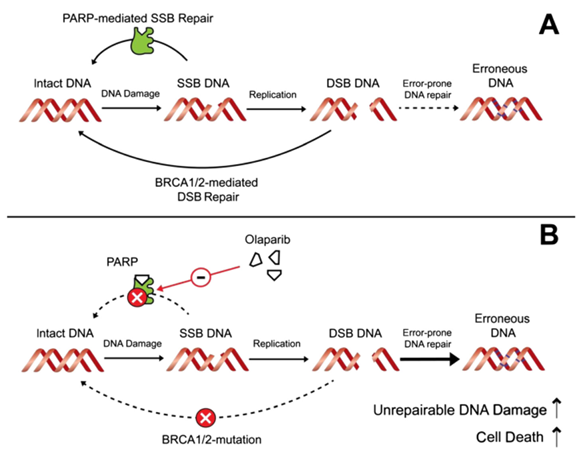
Under normal conditions, single-strand breaks (SSBs) in DNA are repaired by an error-free, poly(ADP-ribose) polymerase
(PARP)-mediated mechanism. When SSBs are present in the DNA during replication, double-strand breaks (DSBs) can
occur (Figure 1A). These are preferentially repaired by the BRCA 1/2-mediated homologousrecombination thatistypically
regarded as an error-free repair mechanism, although recent reports suggest that it can be mutagenic.
Olaparib inhibits the PARP-mediated error-free repair of SSB, resulting in synthetic lethality in BRCA-associated
cancer cells as DNA is then repaired with more error-prone repair mechanisms– single-strand annealing and non
homologous end joining. These alternative repair mechanisms are overwhelmed in the presence of large amounts
of DNA damage– e.g. after treatment with genotoxic agents (Figure 1B). This triggers a build-up of DSBs, erroneous
DNA and, ultimately, cell death[1].
Previous studies have demonstrated that defects in the DNA damage repair response can enhance tumour cell sensitivity to chemotherapy. The Nijmegen breakage syndrome 1 gene (NBS1), encoding a Nibulin homologue protein, constitutes a key component of the MRE11-RAD50-NBN complex (MRN complex) involved in the repair of DNA double-strand breaks (DSBs). Research has revealed that NBS1 deficiency leads to defects in homologous recombination repair. Suppressing NBS1 expression activates the CHK1 and CyclinB signalling pathways, inducing cell cycle arrest and enhancing the sensitivity of ovarian cancer cells to olaparib in both in vitro and in vivo experiments. Ovarian cancer cells lacking NBS1 tend to maintain sensitivity to chemotherapeutic agents by activating cell cycle checkpoints. Consequently, inhibiting NBS1 upregulates CyclinB expression, thereby enhancing ovarian cancer cells' sensitivity to olaparib[2].
Clinical application
Maintenance treatment with olaparib improved median progression-free survival in a Phase II trial with patients with
platinum-sensitive BRCA-mutated serous epithelial ovarian, fallopian tube or primary peritoneal cancer (11.2 months vs.
4.3 months for placebo). However, no significant difference in overall survival was observed between the treatments.
Maintenancetreatmentwitholaparibshouldbeginnolaterthan8weeksafterthefinaldoseofplatinumtherapy.Patients
are directed to take 400 mgorally twice daily, at least 1 hour after food. It is recommended that treatment with olaparib is
stopped upon disease progression.
Adverse effects
Olaparib monotherapy is generally well tolerated, with adverse effects generally of mild-to-moderate severity. Treatment
discontinuation was not frequent in clinical trials. The most commonly observed side effects (>10%) are fatigue, nausea,
vomiting, diarrhoea, dyspepsia, headache, altered taste, decreased appetite and dizziness. An increase in serum creatinine
was also observed in >10% of patients. Haematological toxicity (anaemia, neutropenia, thrombocytopenia and lym
phopenia) has been reported (>10%) in patients treated with olaparib, therefore, patients should not start treatment with
olaparib until they have recovered from any previous chemotherapy-related haematological abnormalities. Regular blood
counts are advised as a risk management strategy. Uncommon serious adverse effects included myelodysplastic syn
drome, acute myeloid leukaemia and pneumonitis.
References
[1] S. C. GOULOOZE R. R A F Cohen. Olaparib[J]. British journal of clinical pharmacology, 2015, 81 1: 171-173. DOI:10.1111/bcp.12761.
[1] AILING ZHONG. Suppression of NBS1 Upregulates CyclinB to Induce Olaparib Sensitivity in Ovarian Cancer.[J]. Technology in Cancer Research & Treatment, 2024, 23: 15330338231212085. DOI:10.1177/15330338231212085.
You may like
Related articles And Qustion
See also
Lastest Price from Olaparib manufacturers

US $5.00-0.50/KG2025-06-05
- CAS:
- 763113-22-0
- Min. Order:
- 1KG
- Purity:
- 99% hplc
- Supply Ability:
- 500TONS

US $1.00/g2025-04-21
- CAS:
- 763113-22-0
- Min. Order:
- 1g
- Purity:
- 99%
- Supply Ability:
- 100kg