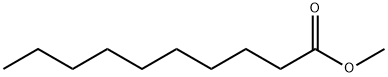
Methyl Decanoate as Biodiesel Surrogate
Methyl decanoate, derived from decanoic acid, has garnered attention as a surrogate for single-component biodiesel. Another noteworthy component, Methyl Caprate, serves as an unsaturated biodiesel fuel surrogate and finds utility as an additive in flavors and fragrances. Exploration into the behavior of methyl decanoate under various conditions has yielded insights. In one study, its thermal decomposition was scrutinized within a jet-stirred reactor operating at temperatures spanning from 773K to 1123K. Furthermore, the oxidation of this compound was investigated within a similar reactor, covering a temperature range of 500K to 1100K. Methyl decanoate is identifiable in diverse sources such as parmesan cheese, sparkling wine, and banana. It stands as a substantial methyl ester, functioning as an effective surrogate for biodiesels.

Study of the thermal decomposition of methyl decanoate
Fuel thermal decomposition reactions are of importance in kinetic studies as the interpretation of experimental phenomena is much simpler than in the case of oxidation reactions for the analysis of the primary reaction pathways of a reactant. Amongst new transportation fuels, the interest in biodiesel is increasing because of their use along with conventional diesel fuels in internal combustion engine. This paper presents new detailed experimental results of the thermal decomposition of methyl decanoate in a jet-stirred reactor. Methyl decanoate (C11H22O2) was chosen because it has been proposed as a possible surrogate for large species found in biodiesel and because a study of the oxidation of this compound was already performed in a jet-stirred reactor. These new results were compared with previous ones obtained for n-dodecane, an n-alkane of similar size, in order to highlight the similarities and the differences between the two types of molecules. A detailed kinetic model for the thermal decomposition of methyl decanoate was generated using the software EXGAS, which was recently used to simulate the oxidation of saturated methyl esters. Computed and experimental data were then compared showing a good agreement.[1]
The feed of liquid methyl decanoate (provided by SAFC, purity: 99+) was controlled thanks a liquid mass flow controller provided by Bronkhorst. This device was associated to an evaporator (also provided by Bronkhorst) in which methyl decanoate was mixed with the carrier gas (helium IC, provided by Messer) and heated to 473 K. Helium flow rate was controlled by a Tylan gas mass flow controller. Helium was used to avoid the condensation of the carrier gas during the sampling at liquid nitrogen temperature at the outlet of the reactor. The pressure in the reactor was maintained constant at 800 Torr (106.6 kPa) using a valve located downstream of the reactor (a pressure of 800 Torr was required because of the pressure drops in the sampling valves of online gas chromatographs). The reaction pathways from methyl decanoate to carbon monoxide and carbon dioxide were investigated from a rate of production analysis at a temperature of 900 K. Percentages are related to the consumption of the reactant (as an example, 1.13% between R43 and R21 means that this reaction corresponds to 1.13% of the consumption of the initial reactan. Toluene is fairly well reproduced by the model, meaning that the concentrations of the phenyl and methyl radicals are likely well predicted.
These new results were compared with previous ones about the pyrolysis of n-dodecane, a linear alkane of similar size. This comparative study showed that both molecules have about the same reactivity, methyl decanoate being slightly more stable than n-dodecane. As far as products are concerned, the main difference is the formation of specific species due to the presence of the ester function in methyl decanoate: unsaturated methyl esters and oxygenated compounds. A model for the thermal decomposition of methyl decanoate was generated using the software EXGAS. The model reproduces fairly well the conversion of the reactant and the mole fractions of the reaction products. The kinetic analysis showed that retro-ene reactions play an important role in the consumption of olefins and esters with one double bond at the extremity of the chain. The model does not account for the evolution of the CO/CO2 ratio at low conversion and for the consumption of small unsaturated methyl esters.
References
[1]Herbinet O, Glaude PA, Warth V, Battin-Leclerc F. Experimental and modeling study of the thermal decomposition of methyl decanoate. Combust Flame. 2011 Jul;158(7):1288-1300. doi: 10.1016/j.combustflame.2010.11.009. PMID: 23710078; PMCID: PMC3661903.
You may like
See also
Lastest Price from Methyl decanoate manufacturers

US $0.00/KG2025-04-21
- CAS:
- 110-42-9
- Min. Order:
- 25KG
- Purity:
- 98%min
- Supply Ability:
- 30tons/month

US $0.00-0.00/KG2025-04-15
- CAS:
- 110-42-9
- Min. Order:
- 25KG
- Purity:
- 99%
- Supply Ability:
- 500000kg