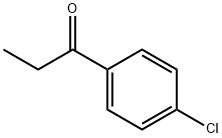
Multifaceted Transformations of 4'-Chloropropiophenone: From Yeast-Mediated Asymmetric Bioreduction to Chemoselective Multiphase Catalytic Hydrodehalogenation
Abstract
4'-Chloropropiophenone can serves as a versatile aromatic ketone and a pivotal synthetic intermediate in both organic and medicinal chemistry. It is a valuable molecular scaffold that demonstrates how precise adjustments in the reaction environment—such as ligand effects, pH, and phase-transfer agents—can dictate competitive pathways between C–Cl bond cleavage, carbonyl hydrogenation, and aromatic ring reduction. It plays an important role in multifaceted chemical transformations under biocatalytic and catalytic regimes, and serves as a model substrate for investigating reaction selectivity.
Biocatalytic Asymmetric Reduction and Antifungal Applications
4'-Chloropropiophenone serves as a versatile substrate for fermentation using baker’s yeast, which is widely recognized for its natural ability to reductively transform diverse ketones into optically active secondary alcohols. This process typically yields a predominantly (S)-configuration product, strictly following the stereochemical logic of Prelog’s rule. Through this specific bioreduction, (S)-(−)-1-(4’-chlorophenyl) propanol is successfully prepared. The resulting compound features a molecular skeleton remarkably similar to various bioactive molecules that demonstrate potent fungicidal activity against Botrytis cinerea, a destructive grey mould known for causing extensive damage to many economically vital crops. Furthermore, the optimal balance between S-enantioselectivity and chemical conversion for this substrate was achieved by implementing a preliminary heat treatment of the yeast cells while utilizing allyl alcohol as a specialized metabolic inhibitor.
A Pivotal Synthetic Intermediate for Bioactive Molecules
4′-Chloropropiophenone is an important aromatic ketone employed as a synthetic intermediate in both organic and medicinal chemistry. It is an effective substrate for diverse functionalization strategies for its reactive carbonyl group and a para-chloro-substituted aromatic ring. In addition, 4′-chloropropiophenone can be utilized as a key intermediate for the construction of biologically active molecules like rimonabant derivatives, because it enables regioselective carbon–carbon bond formation. In addition, 4′-chloropropiophenone commonly participates in reactions for involving enolate formation at the α-position of the ketone. The chloro substituent further facilitates downstream transformations through cross-coupling or substitution reactions. [2]
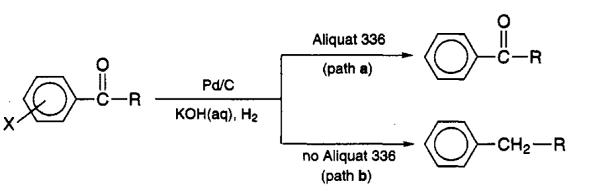
Chemoselective Hydrodehalogenation of 4′-Chloropropiophenone in Biphasic Systems
Early studies reported that 4′-chloropropiophenone undergoes highly chemoselective catalytic transformations under biphasic reaction conditions. When the reaction is carried out in a two-phase system composed of an apolar organic solvent and 50% aqueous KOH at 50 °C, catalytic hydrogenation over Pd/C under atmospheric H₂ in the presence of the phase-transfer catalyst Aliquat 336 (methyltricaprylammonium chloride) leads to selective hydrodehalogenation of the aromatic ring. Only the dehalogenation takes place while the carbonyl group is kept unchanged (path a). In contrast, when operating without Aliquat 336 the product is the corresponding alkyl benzene (path b) hydrocarbons. [3]
pH Effect on the Chemoselectivity and Mechanistic Investigation
4'-Chloropropiophenone was subjected to mild catalytic hydrodehalogenation (p[H2]= 1 atm, T= 50 °C) in a multiphase system consisting of isooctane and an alkaline aqueous phase, in the presence of onium salts as phase-transfer agents. The chemoselectivity of the reaction was sharply influenced by the KOH concentration of the aqueous phase: thus, the formation of 1-phenyl-1-propanol was favored in a highly alkaline pH range (>13.5 by 50% to 5% KOHaq). Instead, up to 95% selectivity toward phenyl ethyl ketone was observed if the pH was controlled (8 < pH < 13) by adjusting the KOHaq concentration in the range between 5 and 1%. Further investigation of the pH effect indicated that an acidic aqueous phase directed the reaction chemoselectivity toward the formation of products arising from aromatic ring reduction (i.e., cyclohexyl ethyl ketone and 1-cyclohexyl-1-propanol) rather than from carbonyl hydrogenation or hydrogenolysis. Also aliphatic amines, used in place of the PT onium salt, afforded good selectivities toward phenyl ethyl ketone, although not as high as the ones obtained with Aliquat 336. [4]
Reference:
[1] Bustillo, A. J., Aleu, J., Hernández-Galán, R., & Collado, I. G. (2002). Biocatalytically assisted preparation of antifungal chlorophenylpropanols. Tetrahedron: Asymmetry, 13(15), 1681-1686.
[2] Donohue, S. R., Halldin, C., & Pike, V. W. (2008). A facile and regioselective synthesis of rimonabant through an enamine-directed 1, 3-dipolar cycloaddition. Tetrahedron letters, 49(17), 2789-2791.
[3] Marques, C. A., Selva, M., & Tundo, P. (1995). Facile hydrodehalogenation with H2 and Pd/C catalyst under multiphase conditions. 3. Selective removal of halogen from functionalized aryl ketones. 4. Aryl halide-promoted reduction of benzyl alcohols to alkanes. The Journal of Organic Chemistry, 60(8), 2430-2435.
[4] Perosa, A., Selva, M., & Tundo, P. (1999). Hydrodehalogenation of Halogenated Aryl Ketones under Multiphase Conditions. 6. pH Effect on the Chemoselectivity and Preliminary Mechanistic Investigation. The Journal of Organic Chemistry, 64(11), 3934-3939.
Related articles And Qustion
Lastest Price from 4'-Chloropropiophenone manufacturers

US $0.00/kg2026-03-02
- CAS:
- 6285-05-8
- Min. Order:
- 1kg
- Purity:
- 99%
- Supply Ability:
- 99999

US $10.00-7.00/KG2026-01-16
- CAS:
- 6285-05-8
- Min. Order:
- 1KG
- Purity:
- 99%
- Supply Ability:
- 100 mt