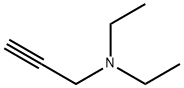
N,N-Diethylpropargylamine: Fine Chemical & Functional Monomer
N,N-Diethylpropargylamine is known for its excellent performance and leveling effect. The compound is insoluble in water and is typically used after reacting with acid to form a quaternary ammonium salt before use. It is also utilized in the fine chemical industry and has applications in pharmaceutical and pesticide intermediates. The synthesis of N,N-Diethylpropargylamine can be achieved through two methods: the atmospheric low-temperature alkalization method and the high-pressure catalysis method. The atmospheric low-temperature alkalization method involves two steps: first, propionic alcohol reacts with phosphorus trichloride to generate propine chlorine under cold conditions, and second, the propine chlorine is reacted with diethylamine under low-temperature alkali conditions to produce N,N-Diethylpropargylamine.

Organometallic polymers from N,N-diethylpropargylamine
Polymers with m-electron delocalised systems have been largely investigated because their conductivity increases by many orders of magnitude by doping. More recently, one-dimensional conjugated polymers were considered of interest because of their non-linear optical properties (NLO). Conducting polymers have been used in the fabrication of light-emitting diodes (LEDs)with different luminescence emission, depending on the chemical structure of the polymer. Organometallic polymers, containing transition metal centers in the conjugated chain, have been synthesised in view of their liquid crystalline behaviour5.6 and of their NLO properties2.Polymers in which metal centers were coordinated to t-systems of the polymeric chain were also prepared, showing conductivity higher than the pristine materials. We found that, by increasing the amount of transition metal complex, the yield of organometallic polymers, in which the metal centers are in part inserted in the main chain and in part coordinated to the pending groups or to the double bonds of the chain, increases. In this paper we will discuss the reactivity of N,N-Diethylpropargylamine (DEPA)with the [Ni(NCS)2(PPh3)2] complex. The present results will be compared with those of our previous investigations. DEPA reacts with [Ni(NCS)2(PPh3)2] giving organometallic polymers. Analogous behaviour was found for N,N-Diethylpropargylamine in reactions with the same complex and with the [Pd(C=CCH2NMe2)2(PPh3)2] complex, which gives polymers containing increasing amounts of metal by increasing the complex/monomer ratio. A different sub- stituted acetylene propargyl alcohol (H-C=C-CH2OH) reacts in a similar way with the related bis-acetylide [Pd(C=CCH2OH)2(PPh3)2] complex.[1]
In this investigation we have found that the polymers obtained by N,N-Diethylpropargylamine contain Ni atoms bonding at least one OPPh3 or PPh3 molecule and about two NCS groups. The Ni2p3 XPS signal of both PDMPA and PDEPA exhibit shake up satellites, which indicate the formation of paramagnetic Ni complexes. The shake up satellites are associated to tetrahedral or octahedral arrangements around the Ni atoms, the latter arrangement indicating crosslinking between polymer chains. Our investigations have shown that monosubstituted acetylenes, containing functionalised pending groups, can be polymerised in the presence of some metal complexes, which act as polymerisation catalysts but react also with the functionalised groups of the monomer, giving organo- metallic polymers. The chemical structure and the nature of the Ni coordination sites have been elucidated by XPS studies. In the polymers of N,N-Diethylpropargylamine, paramagnetic Ni(II)is found in both tetrahedral and octahedral configurations. The octahedral Ni(II)is bonded to two NCS, one OPPh3, and to NEt2 ligands of the polymer chain pending groups. Interaction of Ni with the polymer C=C bonds may also occur. Electrical measurements indicate the possible use of these materials in the preparation of gas sensors. Doped PDEPA are shown to be sensitive to humidity variations in the 50- 90%relative humidity range, giving reproducible conductivity values in cyclic measurements. The doped polymers of N,N-Diethylpropargylamine is poorly soluble. The aim of future investigations will be to prepare new organometallic polymers with similar properties and increased solubility, in order to obtain films which could be used for electronic applications.
Mixed-mode chromatography stationary phase for protein separation
In this study, N,N-Diethylpropargylamine was covalently bonded to the azide-silica by a click-reaction to obtain a novel dual-function mixed-mode chromatography stationary phase for protein separation with a ligand containing tertiary amine and two ethyl groups capable of electrostatic and hydrophobic interaction functionalities, which can display hydrophobic interaction chromatography character in a high-salt-concentration mobile phase and weak anion exchange character in a low-salt-concentration mobile phase employed for protein separation. Based on this MMC column involving N,N-Diethylpropargylamine, a new 2DLC technology was developed using only a single MMC column, which is very important not only to the development of new 2DLCtechnology with a single column, but also to recombinant protein drug production for saving column expense and simplifying the process in biotechnology. Nine kinds of tested proteins can be separated completely using the developed method within 2.0 h.
References
[1]. Furlani . (1998). Organometallic polymers from N,N-diethylpropargylamine containing Ni coordination centers. Polymer, 39 15, Pages 3341-3348. https://doi.org/10.1016/S0032-3861(97)10262-2
[2]Zhao, Kailou et al. “Preparation of a weak anion exchange/hydrophobic interaction dual-function mixed-mode chromatography stationary phase for protein separation using click chemistry.” Journal of separation science vol. 38,5 (2015): 703-10. doi:10.1002/jssc.201401020
You may like
Lastest Price from N,N-Diethylpropargylamine manufacturers

US $45.00/kg2025-04-21
- CAS:
- 4079-68-9
- Min. Order:
- 1kg
- Purity:
- 99%
- Supply Ability:
- 20 tons

US $150.00/kg2025-04-21
- CAS:
- 4079-68-9
- Min. Order:
- 1kg
- Purity:
- 99%
- Supply Ability:
- 500kg