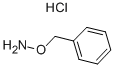
O-Benzylhydroxylamine Hydrochloride: Synthesis & Applications
O-Benzylhydroxylamine hydrochloride and its derivatives represent a class of organic compounds with significant utility in synthetic chemistry and drug discovery. The core structure, characterized by a benzyloxy group attached to a nitrogen atom, provides a versatile scaffold for the synthesis of a wide array of more complex molecules. Initially explored within the realm of classical organic synthesis, the applications of these compounds have expanded dramatically. They are now recognized as crucial intermediates in the preparation of pharmaceuticals, agrochemicals, and fine chemicals. In medicinal chemistry, the O-Benzylhydroxylamine hydrochloride moiety is a key building block for creating oximes and hydroxamic acids, functionalities present in numerous biologically active agents. Furthermore, these derivatives have emerged as potent modulators of biological pathways, most notably as inhibitors of the enzyme Indoleamine 2,3-dioxygenase-1 (IDO1), a critical target in cancer immunotherapy.

Synthesis of oximes derived from O-Benzylhydroxylamine hydrochloride
The spread of multidrug-resistant bacteria has drastically impaired the efficacy of antibiotics, limiting their clinical use. In September 2010, a report of the emergence of Gram-negative Enterobacteriaceae with resistance to carbapenem conferred by New Delhi metallo-β-lactamase 1 (NDM-1) drew global attention. To prevent these serious medical threats, the elaboration of new types of antibacterial agents and improvement of drugs in current or prior use are very important tasks. Several kinds of compounds have been designed and screened in enzyme assays to generate leads that were co-crystallized with various pathogenic FabH proteins and subsequently optimized using structure-guided drug design methods. For this study, we designed and synthesized a new series of oxime derivatives with a methyleneaminoxy group (CNO) derived from O-Benzylhydroxylamine hydrochloride and primary benzaldehydes or salicylaldehydes, and then studied their antimicrobial activities and inhibitory activities toward E. coli FabH. Docking simulations were performed using the X-ray crystallographic structure of E. coli FabH in complex with the most active inhibitor, to explore the binding mode of the compound at the active site. This series of compounds was found to be more efficient than most of the previously reported FabH inhibitors, and their structures are also different from most reported inhibitors. O-Benzylhydroxylamine hydrochloride was prepared by reaction of benzyl halide and N-hydroxyphthalimide in dimethyl sulfoxide (DMSO) in the presence of sodium acetate trihydrate followed by hydrolysis of the imidic group by treatment with hydrazine hydrate.[1]
A series of novel oxime derivatives generated by combining substituted benzaldehydes and salicylaldehydes with substituted O-Benzylhydroxylamine hydrochlorides were assayed for their antibacterial activities against E. coli, P. aeruginosa, P. fluorescens, B. subtilis, S. aureus, and E. faecalis. 3-((2,4-dichlorobenzyloxyimino)methyl)benzaldehyde O-2,4-dichlorobenzyl oxime, showed the best antibacterial activity with MICs of 3.13–6.25 μg mL−1 against the strains tested, and also exhibited the highest E. coli FabH inhibitory activity, with IC50 1.7 μM. The introduction of hydrophobic and electron-withdrawing groups was conducive to the antibacterial and FabH inhibitory activities. Docking simulations were performed to position 3-((2,4-dichlorobenzyloxyimino)methyl)benzaldehyde O-2,4-dichlorobenzyl oxime into the E. coli FabH active site to determine the probable binding conformation.
O-Benzylhydroxylamine hydrochloride for analysis of short-chain fatty acids
Short-chain fatty acids (SCFAs) are metabolites derived from gut microbiota and implicated in host homeostasis. Hence, the profiling SCFAs from biological samples plays an important role in revealing the interaction between gut microbiota and pathogens. Previous studies, liquid chromatography–tandem mass spectrometry (LC–MS/MS) combined with various derivatization strategies have been performed to obtain the SCFA profiles from biological samples. However, it is poor evidence to compare these derivatization regents and conditions. Thus, we present the evaluation of three major derivatization reagents, namely 3-nitrophenylhydrazine (3-NPH), O-Benzylhydroxylamine hydrochloride (O-BHA), and 2-picolylamine (2-PA), for the analysis of eight SCFAs classified as C2–C5 isomers using LC–MS/MS. First, in a reversed-phase LC separation, 3-NPH showed good retention capacity. Although O-BHA derivatization showed higher sensitivity and good retention capacity than 2-PA, only 2-PA derivatization could successfully separate eight SCFAs. The matrix effects in human serum ranged 77.1–99.0% (RSD ≤ 3.4%, n = 6) for 3-NPH derivatives, 91.0–94.6% (RSD ≤ 5.4%, n = 6) for O-Benzylhydroxylamine hydrochloride derivatives, 81.6–99.5% (RSD ≤ 8.0%, n = 6) for 2-PA derivatives. These compared results showed each characteristic of 3-NPH, O-BHA, and 2-PA for SCFA derivatization based on LC–MS/MS approaches.[2]
References
[1]Luo, Yin et al. “Synthesis and antimicrobial activities of oximes derived from O-benzylhydroxylamine as FabH inhibitors.” ChemMedChem vol. 7,9 (2012): 1587-93. doi:10.1002/cmdc.201200225
[2]Nagatomo, Ryosuke et al. “Comparison of 3-nitrophenylhydrazine, O-benzyl hydroxylamine, and 2-picolylamine derivatizations for analysis of short-chain fatty acids through liquid chromatography coupled with tandem mass spectrometry.” Analytical sciences : the international journal of the Japan Society for Analytical Chemistry vol. 40,5 (2024): 843-851. doi:10.1007/s44211-023-00474-7
See also
Lastest Price from O-Benzylhydroxylamine hydrochloride manufacturers

US $0.00/G/KG2025-04-23
- CAS:
- 2687-43-6
- Min. Order:
- 1G/KG
- Purity:
- 99%
- Supply Ability:
- 100MT

US $0.00/G/KG2025-04-23
- CAS:
- 2687-43-6
- Min. Order:
- 1G/KG
- Purity:
- 99%
- Supply Ability:
- 100MT