Pharmacological Action and Directions for Use of Besifloxacin hydrochloride
Besifloxacin hydrochloride is an anti-infective pharmaceutical molecule, commonly formulated as an ophthalmic solution, and is frequently used to treat bacterial conjunctivitis caused by susceptible pathogens. As an 8‑chloro‑fluoroquinolone bearing an N‑1 cyclopropyl group, besifloxacin hydrochloride inhibits both bacterial DNA gyrase and topoisomerase IV, and exhibits activity against both Gram‑positive and Gram‑negative bacteria.

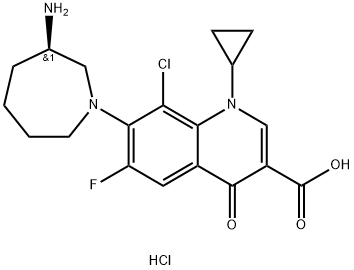
Figure1: Picture of Besifloxacin Hydrochloride
Overview
Besifloxacin Hydrochloride is a fourth generation fluoroquinolone antibiotic. Besifloxacin Hydrochloride is a DNA gyrase and topoisomerase IV inhibitor. Besifloxacin Hydrochloride has broad-spectrum antibacterial activity, it is effective against Gram-negative and Gram-positive aerobic and anaerobic strains and reduces the incidence of drug resistance. Besifloxacin Hydrochloride has anti-inflammatory activity. Besifloxacin Hydrochloride can be used in bacterial conjunctivitis research.
Pharmacological Action
In vitro studies show that the incidence of resistance to Besifloxacin hydrochloride in Staphylococcus aureus is less than 3.3×10⁻¹⁰, and in drug-resistant Streptococcus pneumoniae it is less than 7×10⁻¹⁰. Besifloxacin hydrochloride exhibits activity in vitro and in clinical trials of conjunctival infections against most of the following bacteria, including: Corynebacterium group G, Corynebacterium pseudodiphtheriticum, Corynebacterium striatum, Haemophilus influenzae, Moraxella catarrhalis, Staphylococcus aureus, Staphylococcus epidermidis, Staphylococcus haemolyticus, Staphylococcus lugdunensis, Streptococcus mitis group, Streptococcus oralis, Streptococcus pneumoniae, and Streptococcus salivarius isolates.
Synthesis
The present invention relates to a method of antimicrobial hydrochloride Besifloxacin a fluoroquinolone. The method of 1-cyclopropyl-6,7-difluoro-8-chloro-1,4-dihydro-4-oxo - quinoline-3-carboxylic acid -O3, O4- bonded boron diacetate ester and ( R) -3- trityl-amino - hexahydro-azepine, or a salt thereof as raw materials, condensation Besifloxacin Hydrochloride was prepared in three steps. The method reliable, efficient and suitable for industrial production. [1]
In Vivo Experiments
Besifloxacin hydrochloride demonstrates high antibacterial activity against clinical isolates of Staphylococcus aureus, with a minimum inhibitory concentration (MIC) range of 0.015–8 μg/mL. Studies indicate that as the number of mutations in the QRDR region of the bacterial strains increases, the rise in MIC for besifloxacin hydrochloride is significantly smaller compared to other fluoroquinolones (such as ciprofloxacin and levofloxacin), suggesting it retains stronger efficacy against resistant strains, particularly those resistant to ciprofloxacin. In vivo experiments show that besifloxacin hydrochloride (6 mg/mL, administered topically with 19 instillations within 8 hours) significantly reduces the clinical severity of infections caused by methicillin-resistant Staphylococcus aureus (MRSA) and effectively decreases the bacterial load of MRSA in the cornea. The minimum inhibitory concentration of besifloxacin hydrochloride (1 μg/mL) is 8-fold lower than that of other fluoroquinolone drugs. [2]
Application
Besifloxacin hydrochloride is a fourth-generation fluoroquinolone antibiotic. In vitro, besifloxacin inhibits the production of pro-inflammatory cytokines. Besifloxacin hydrochloride is a novel 8‑chloro‑fluoroquinolone with potent bactericidal activity against common drug‑resistant pathogens.
Directions for Use
Besifloxacin hydrochloride is intended for topical ophthalmic use only and should not be administered via subconjunctival injection or by direct injection into the anterior chamber. Hands should be washed before using this product. To avoid contamination, do not touch the dropper tip to the eye, fingers, or any other surface. Do not rinse the dropper with water. After each use, tighten the cap securely. If another ophthalmic medication (e.g., eye drops or ointment) is being used, wait at least 5 minutes before administering the other medication, and besifloxacin hydrochloride should be used prior to other eye medications. Do not share this product with others, and avoid wearing contact lenses during treatment. Besifloxacin hydrochloride is only for treating the patient’s current diagnosed condition and should not be used to treat other infections; additional infections require separate treatment. If a dose is missed, administer it as soon as possible unless it is nearly time for the next dose; do not double the dose. Continue to complete the full course of treatment even after symptoms improve, as stopping too early may lead to recurrence.
Ocular Irritation
Objective: To observe reversible inflammatory reactions and other potential irritant responses on the anterior ocular surface after administration of Besifloxacin hydrochloride eye drops in rabbit eyes. Method: Four New Zealand white rabbits that passed the fluorescein sodium eye examination were used. Each rabbit received continuous self‑controlled bilateral dosing for 7 days, with the left eye administered the test product and the right eye given an equal volume of blank vehicle. Ocular examinations were performed daily before dosing, as well as at 1, 2, 4, 24, 48, and 72 hours after the last administration. At the end of the study, the eyeballs were collected for histopathological examination. Results: Continuous administration of the undiluted Besifloxacin hydrochloride eye drops for 7 days did not cause significant irritation to rabbit eyes. Conclusion: Besifloxacin hydrochloride eye drops are non‑irritating to rabbit eyes. [3]
References
[1] Chen Z X, Xia Z J, Jiang L, et al. Preparation method of besifloxacin hydrochloride[J]. 2017.
[2] Sanfilippo C M, Hesje C K, Haas W, et al. Topoisomerase mutations that are associated with high?level resistance to earlier fluoroquinolones in Staphylococcus aureus have less effect on the antibacterial activity of besifloxacin[J]. Chemotherapy, 2012, 57: 363?371.
[3] Qian C, Sun B H, Chen Z X, et al. Ocular irritation of besifloxacin hydrochloride[J]. West China Journal of Pharmaceutical Sciences, 2013, 28(2).
You may like
Related articles And Qustion
See also
Lastest Price from Besifloxacin hydrochloride manufacturers
US $0.00/kg2025-11-21
- CAS:
- 405165-61-9
- Min. Order:
- 1kg
- Purity:
- 98%
- Supply Ability:
- Customise

US $0.00/Kg/Drum2025-04-21
- CAS:
- 405165-61-9
- Min. Order:
- 1KG
- Purity:
- 99%
- Supply Ability:
- 500kg