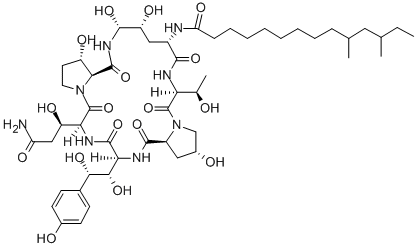
Pneumocandin B0: Echinocandin Antifungal & Fermentation Production
Pneumocandin B0 is a naturally occurring glareacin-type substance belonging to a class of lipopeptides, renowned for its role in inhibiting fungal cell wall synthesis. This compound is extracted from the fermentation process of the fungus Glarea lozoyensis and serves as a key starting material for the synthesis of more effective antifungal drugs, such as caspofungin.

Pneumocandin B0 in extractive batch fermentation of Glarea lozoyensis
Pneumocandin B0, an antifungal agent produced by G. lozoyensis, is a lipohexapeptide of the echinocandin family that inhibits fungal 1, 3-β-glucan synthase. In 1987, it was discovered among various minor components of pneumocandin fermentations. This minor component was chosen as natural starting material for the synthesis of caspofungin acetate (CANCIDAS®). G. lozoyensis was recognized as a novel fungus through DNA fingerprinting and rDNA sequence analysis. Because of the difficulty in used in traditional protoplast transformation techniques with this fungus, Agrobacterium-mediated transformation was developed as a simple and efficient in gene replacement method. Extractive fermentation technology has been successfully applied as an effective method for improving the extraction of fungal intracellular products. With the addition of extractive agents in the fermentation broth, the micellare aqueous solution can separate into two phases, where one is a dilute phase (aqueous solution) while the other is a coacervate phase (extractant-rich phase). Intracellular products are released from the intracellular to the extracellular and progressively extracted into the coacervate phase. Thus the product is continuously extracted into the nonaqueous solvent phase and the fungal cells continuously produce hydrophobic metabolites. In this work, we investigated the influence of different extractants on the pneumocandin B0 yield and explored the possible reason for improved pneumocandin B0 production in the extraction system. Moreover, the response of the putative trans-membrane secretion model of pneumocandin B0 in extractive fermentation was established accordingly.[1]
SDS is an amphiphilic compound that has both water and oil solubility, and its structure is similar to the structure of phospholipids molecules in the cell membrane. Consequently, the added SDS could form a complex with membrane phospholipids to form mixed micelles which would greatly alter the structure of the cell membrane and improve its permeability, making the membranes more conducive to the export of intracellular pneumocandin B0. The fatty acid composition of the cell membrane has a great influence on permeability. The saturated fatty acids in the cell membranes are linear, with tight inter chain arrangements and large interactions, resulting in low penetrability of the membrane. Unsaturated fatty acids are bent, making it difficult for the two fatty acid chains of phospholipids to align close to each other, resulting in increased penetrability of the membrane. Based on the results, we postulated a putative trans-membrane release model of pneumocandin B0 in extractive fermentation (Fig. 4). With the addition of SDS, the increased incorporation of unsaturated fatty acids in the cell membrane and mixed micelles improved the membrane permeability, facilitating the release of intracellular pneumocandin B0 and allowing the intracellular synthesis of new pneumocandin B0. Furthermore, the reduction of hyphal and pellet diameters facilitates higher dissolved oxygen and more efficient exchange of nutrients to cells of G. lozoyensis.
Enhancement of Pneumocandin B0 Production in Glarea lozoyensis
The production of pneumocandin B0 is limited by feedback inhibition. Here, low-temperature adaptive laboratory evolution (ALE) was used to improve the production capacity of Glarea lozoyensis by enhancing its membrane permeability. After 50 cycles of ALE, the pneumocandin B0 production of the endpoint strain (ALE50) reached 2131 g/L, which was 32% higher than the starting strain (ALE0). ALE50 showed a changed fatty acid composition of the cell membrane, which-+h increased its permeability by 14%, which in turn increased the secretion ratio threefold. Furthermore, ALE50 showed increased intracellular proline and acetyl-CoA concentrations, superoxide dismutase (SOD), and catalase (CAT) activity, as well as total antioxidant capacity. The slight biomass decrease in ALE50 was accompanied by decreased isocitrate dehydrogenase (ICDH) and glucose-6-phosphate dehydrogenase (G6PDH) activity. Finally, a putative model of the accumulation and secretion of pneumocandin B0 in ALE50 was established. ALE is a promising method to release intracellular feedback inhibition.[2]
References
[1]Yuan K, Huang B, Qin T, Song P, Zhang K, Ji X, Ren L, Zhang S, Huang H. Effect of SDS on release of intracellular pneumocandin B0 in extractive batch fermentation of Glarea lozoyensis. Appl Microbiol Biotechnol. 2019 Aug;103(15):6061-6069. doi: 10.1007/s00253-019-09920-x. Epub 2019 Jun 3. PMID: 31161390; PMCID: PMC6616208.
[2]Song P, Zhang K, Zhang S, Huang BQ, Ji XJ, Ren LJ, Gao S, Wen JP, Huang H. Enhancement of Pneumocandin B0 Production in Glarea lozoyensis by Low-Temperature Adaptive Laboratory Evolution. Front Microbiol. 2018 Nov 21;9:2788. doi: 10.3389/fmicb.2018.02788. PMID: 30519220; PMCID: PMC6259640.
You may like
Lastest Price from Pneumocandin B0 manufacturers
US $0.00/kg2025-11-21
- CAS:
- 135575-42-7
- Min. Order:
- 1kg
- Purity:
- 98%
- Supply Ability:
- Customise

US $0.00/KG2025-04-21
- CAS:
- 135575-42-7
- Min. Order:
- 1KG
- Purity:
- 95%min
- Supply Ability:
- 50kg