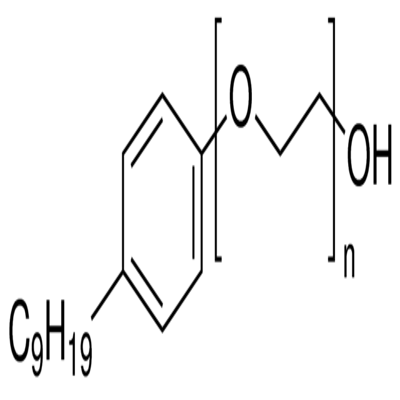
Polyethylene glycol mono-4-nonylphenyl ether: Spermicidal Surfactant & Contraceptive
Polyethylene glycol mono-4-nonylphenyl ether(Nonoxynol-9) is a chemical compound that reduces surface tension (surfactant). It acts on the surface of the sperm and damages its cell membrane, thereby immobilizing, inactivating and killing the sperm. Most nonoxynol-9 products are required to be used before each act of intercourse and remain effective locally, usually for an hour. Some of the nonoxynol-9 products such as the vaginal sponge also work as barrier contraceptives. Polyethylene glycol mono-4-nonylphenyl ether has a higher failure rate than other contraception methods, and does not provide protection against human immunodeficiency virus (HIV) or other sexually transmitted diseases. It should ideally be used along with vaginal diaphragms or male latex condoms for more effective contraception.

Effects of Polyethylene glycol mono-4-nonylphenyl ether on sperm functions
Spermicide is a chemical barrier contraceptive used before intercourse for birth control, which has been used for thousands of years (Duffy & Archer 2018). The basis of spermicidal action is to immobilize or disrupt the plasma membrane of the sperm, so as to avoid sperm–egg interaction (Ingram et al. 2006). Polyethylene glycol mono-4-nonylphenyl ether (N-9) is widely used as an active ingredient in various spermicides. Spermicides containing N-9 do not require a prescription but may be available online or in the drugstore from low to high prices without age restrictions. It may be available from some health centers as well. While other products have been tested and one recently has been approved by the US Food and Drug Administration (FDA), N-9 is the most commonly used agent (Eisenberg 2020, Thomas et al. 2020). Its products constitute large proportions of the spermicides easily accessible in the market at affordable prices (Grimes et al. 2013). It can still be one of the options for women with a low risk of sexually transmitted diseases to prevent pregnancy. Phase I and II clinical trials studying the effects of polyethylene glycol mono-4-nonylphenyl ether focused on the sperm structure and functions. We also want to find the inconstancies in the previous studies on N-9 and try to resolve the conflicts. Additionally, with the help of this review, we hope to provide new insights for further research on N-9. Thus, we conducted the systematic review and meta-analysis of published papers on functional mechanisms of polyethylene glycol mono-4-nonylphenyl ether on sperm structure and functions to update current understanding.[1]
In this systematic review and meta-analysis, we included 11 eligible studies to assess the impacts of polyethylene glycol mono-4-nonylphenyl ether on sperm with the changes in numbers of PR sperm in cervical mucus demonstrated by PCT in in vivo studies as the primary outcome and vanguard sperm penetration distances in in vitro studies as the secondary outcome. The primary result showed that, in cervical mucus, the number of PR sperm per HPF decreased significantly after N-9 treatment when compared with the personal baselines. As one meta-analysis indicates that vanguard sperm penetration distance has low accuracy in evaluating sperm motility compared with sperm count/HPF, this was taken as the secondary outcome. Since it has been confirmed to be effective in killing sperm, more research can be done to explore complementary agents like anti-microbiotics or other agents which can change the vagina environment to achieve the goal of preventing pregnancy more effectively and to confer non-contraceptive benefits such as microbicidal effects. In conclusion, polyethylene glycol mono-4-nonylphenyl ether has several effects on sperm owing to its potency in reducing sperm motility and cervical mucus penetration, as well as other functional competencies.
In Vitro and In Vivo of Polyethylene glycol mono-4-nonylphenyl ether
The current impact of the global AIDS epidemic is staggering, with even greater potential to come. Of the more than 14,000 estimated new HIV infections every day, half occur in young persons between the ages of 15 and 24 years, and teenage girls are disproportionately affected compared with boys. The first microbicidal product to be clinically evaluated contained polyethylene glycol mono-4-nonylphenyl ether (N-9), a nonionic surfactant as the active agent. N-9 has been used at concentrations between 2% and 12% as the active component of spermicides for more than a quarter of a century. The spermicidal action of N-9 is attributed to solubilization and disruption of the spermatozoa plasma membranes, and it is used in the laboratory for extraction of membrane-bound proteins without denaturation. Early studies indicated that N-9 was active against a range of microbes and/or pathogens in vitro, including herpes simplex virus (HSV), cytomegalovirus, Neisseria gonorrhoeae, Trichomonas vaginalis, Treponema pallidum, Gardnerella vaginalis, Bacteroides, and Chlamydia trachomatis. The World Health Organization (WHO)/Contraceptive Research and Development (CONRAD) Technical Consultation on polyethylene glycol mono-4-nonylphenyl ether Report, a panel convened in October 2001, concluded that N-9 should not be used for the purpose of STI or HIV prevention. The wealth of preclinical and clinical data available for N-9 represents an important opportunity to assess the predictive value of this data in relation to the clinical outcome, however.[2]
N-9 was never subjected to the extensive in vitro preclinical evaluation that is now required of all candidates before clinical testing. In the current scientific environment, it is highly unlikely that N-9 would have progressed further than early in vitro evaluation. In vitro, in vivo, and clinical studies have suggested the potential ability of polyethylene glycol mono-4-nonylphenyl ether to exert an influence on inflammatory processes. These findings have started a debate on what is the best method to detect vaginal inflammation in clinical trials of microbicidal candidates. Evaluating cervical lesions has been a routine part of clinical safety assessments for microbicide trials; however, little is known about more subtle changes in the cervical and vaginal epithelium, including induction of subepithelial inflammation and interference with normal host defense mechanisms. The lack of correlation in clinical studies of inflammation, cervical lesions, and symptomology suggests that we need to address this issue further in all future trials. Polyethylene glycol mono-4-nonylphenyl ether has provided us with an invaluable learning experience in microbicide preclinical development. The lessons learned from polyethylene glycol mono-4-nonylphenyl ether studies have and should continue to be in force in the design of protocols to test other candidate microbicides. The most critical aspect of this experience has been to recognize the paramount importance of safety in microbicide development.
References
[1]Xu M, Zhao M, Li RHW, Lin Z, Chung JPW, Li TC, Lee TL, Chan DYL. Effects of nonoxynol-9 (N-9) on sperm functions: systematic review and meta-analysis. Reprod Fertil. 2022 Feb 21;3(1):R19-R33. doi: 10.1530/RAF-21-0024. PMID: 35350652; PMCID: PMC8956826.
[2]Hillier, Sharon L et al. “In vitro and in vivo: the story of nonoxynol 9.” Journal of acquired immune deficiency syndromes (1999) vol. 39,1 (2005): 1-8. doi:10.1097/01.qai.0000159671.25950.74
You may like
See also
Lastest Price from POLYETHYLENE GLYCOL MONO-4-NONYLPHENYL ETHER manufacturers

US $10.00/ASSAYS2025-08-21
- CAS:
- 26027-38-3
- Min. Order:
- 1ASSAYS
- Purity:
- 99%
- Supply Ability:
- 1 ton

US $0.00-0.00/KG2025-08-07
- CAS:
- 26027-38-3
- Min. Order:
- 1KG
- Purity:
- 99%
- Supply Ability:
- 1tons