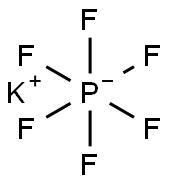
Potassium Hexafluorophosphate: Inorganic Fluoride Salt for Advanced Materials
Potassium Hexafluorophosphate is a water insoluble Potassium source for use in oxygen-sensitive applications, such as metal production. In extremely low concentrations (ppm), fluoride compounds are used in health applications. Fluoride compounds also have significant uses in synthetic organic chemistry. They are commonly also used to alloy metal and for optical deposition. Certain fluoride compounds can be produced at nanoscale and in ultra high purity forms. Potassium Hexafluorophosphate is generally immediately available in most volumes. Ultra high purity and high purity compositions improve both optical quality and usefulness as scientific standards.

Influence of Potassium Hexafluorophosphate on the AM50 Magnesium Alloy
Magnesium is the lightest construction material (1.74 g/mL), but due to its high reactivity and poor mechanical properties, it is practically not used in its pure form. The most frequently added alloying element is aluminum, which ensures high strength, creep resistance, and anticorrosive properties. The most commonly used types of baths in the PEO process include silicate and phosphate electrolytes, which ensure appropriate mechanical and anticorrosive properties of the coatings are produced. Baths combining both components are also used to further optimize the protective properties of the synthesized surface layers. In this study, the influence of the addition of potassium hexafluorophosphate to electrolytes on the structure and anticorrosive properties of PEO coatings produced on the AM50 magnesium alloy has been investigated. To our knowledge, the application of KPF6 to electrolytic baths has not yet been reported. The analysis of the impact of the addition of potassium hexafluorophosphate on the PEO process is interesting because there are reports in the literature that state that the PF6− ion is not hydrolyzed in a strongly alkaline medium (pH > 12), unlike K2ZrF6, K2TiF6, or Na3AlF6. Therefore, the formation of a polyfluorine magnesium salt in the structure of the conversion coating is possible, as we found in the case of the addition of NaBF4 salt. The properties of conversion coatings prepared at the optimal concentration of hexafluorophosphate ions have also been compared with those produced in a bath containing equimolar amounts of fluorine and phosphorus in the form of the most commonly used fluoride and orthophosphate salts.[1]
In the presented study, the PEO coatings produced on the AM50 Mg alloy in alkaline silicate baths with the addition of potassium hexafluorophosphate have been investigated. The results obtained can be summarized as follows: The anticorrosive properties of the obtained coatings increase when the KPF6 concentration is increased to 2.5 g/L. The addition of larger amounts of KPF6 causes damage to the coating (a large increase in its roughness), probably due to the local formation of HF during the PEO process. The addition of potassium hexafluorophosphate allows for better anticorrosive properties of the synthesized coating to be obtained, compared to a mixture of NaF and Na3PO4 with an equimolar content of fluorine and phosphorus. XPS measurements have shown that in coatings obtained in the presence of KPF6, as well as a mixture of NaF and Na3PO4 in the baths, the coating components derived from these additives are the same [MgF2 and Mg3(PO4)2]. Mg(PF6)2 was not present in the formed coatings, which is in contrast to the formation of Mg(BF4)2, when the silicate bath contained NaBF4. The surface morphology of the PEO coatings produced in the potassium hexafluorophosphate-containing baths was more uniform and showed a sponge-like structure, in contrast to commonly reported crater-like structures. The sponge-like structure is similar to bone structure, and in combination with the presence of phosphates, it can increase the biocompatibility and the possibility of self-healing of this coating.
Plating and Stripping Calcium Metal in Potassium Hexafluorophosphate Electrolyte
The ever-increasing energy demand from developing technologies, along with the limited supply of lithium to meet them, has necessitated the need to explore post-lithium-ion battery solutions. An alternate strategy that bypasses the need for elevated temperatures and high overpotentials is the use of a hybrid SEI. The use of mixed cations in the SEI has proven effective at cycling with lithium anodes. Here, we report the development of such a hybrid SEI using potassium hexafluorophosphate (KPF6) salt in a composite solvent of ethylene carbonate (EC), dimethyl carbonate (DMC), and ethyl methyl carbonate (EMC) as it is cycled between calcium metal electrodes. The plating and stripping behavior observed maintained overpotentials below 2 V throughout the course of cycling, while also exhibiting discrete events of potassium and calcium deposition onto the electrodes. All electrochemistry experiments used a 1 M solution of potassium hexafluorophosphate in EC/DMC/EMC. The solvents were first mixed in a 1:1:1 volume ratio for 24 h before being dried for 48 h over molecular sieves. The KPF6 was dried overnight in a vacuum oven at 120 °C before being added to the solvent mixture. The KPF6 solution was stirred for 24 h to allow full dissolution of the salt and was again dried over molecular sieves. Water content of the electrolyte was verified by a Karl Fisher Titrator to be below 50 ppm.[2]
The overpotentials from plating and stripping remain at 1.8 V (vs Ca/Ca2+) for over 20 cycles. Unlike plating and stripping experiments that use Ca(PF6)2 where the overpotentials quickly rise to 5 V, cycling with KPF6 is far more stable. The potassium hexafluorophosphate electrolyte remained stable with the gold electrode up to 4 V, confirming that no side reactions are occurring on the gold electrode. We have shown successful plating and stripping with calcium metal electrodes using a native potassium electrolyte while maintaining overpotentials at 2 V. The SEI formed from the galvanostatic cycling plates and strips both calcium and potassium, as observed with XRD, FTIR, and EDX. The SEI formed on the calcium electrodes is influenced by the two regions of plating and stripping behavior with Ca, CaF2, and P2O5 phases forming at the 0.2 V overpotential and K2P2O5F2 and K phases forming at 1.8 V. The SEI also has a mixture of permanent and transient phases with K and K2P2O5F2 being removed after stripping while CaF2 and P2O5 remain. The uptake of calcium into the potassium hexafluorophosphate electrolyte over the course of cycling transitions the conducting media to a dual ion system that increases the availability of calcium. This work provides demonstration that potassium electrolytes provide similar benefits to the plating and stripping of calcium that has been observed with sodium electrolytes.
Potassium Hexafluorophosphate Enables Stable Lithium-Sulfur Batteries
Uncontrollable dendrite growth and low Coulombic efficiency are the two main obstacles that hinder the application of rechargeable Li metal batteries. Here, an optimized amount of potassium hexafluorophosphate (KPF6, 0.01 M) has been added into the 2 M LiTFSI/ether-based electrolyte to improve the cycling stability of lithium-sulfur (Li-S) batteries. Due to the synergistic effect of self-healing electrostatic shield effect from K+ cations and the LiF-rich solid electrolyte interphases derived from PF6- anions, the potassium hexafluorophosphate additive enables a high Li Coulombic efficiency of 98.8% (1 mA cm-2 of 1 mAh cm-2). The symmetrical Li cell can achieve a stable cycling performance for over 200 cycles under a high Li utilization up to 33.3%. Meanwhile, the polysulfide shuttle has been restrained due to the higher concentration of the LiTFSI in the electrolyte. As a result, the assembled Li-S full cell displays excellent capacity retention with only 0.25% decay per cycle in the final electrolyte. Our work offers a smart approach to improve both the anode and cathode performance by the electrolyte modification of rechargeable Li-S batteries.[3]
References
[1]Florczak Ł, Kościelniak B, Kramek A, Sobkowiak A. The Influence of Potassium Hexafluorophosphate on the Morphology and Anticorrosive Properties of Conversion Coatings Formed on the AM50 Magnesium Alloy by Plasma Electrolytic Oxidation. Materials (Basel). 2023 Dec 9;16(24):7573. doi: 10.3390/ma16247573. PMID: 38138715; PMCID: PMC10744744.
[2]Chando PA, Shellhamer JM, Wall E, He W, Hosein ID. Plating and Stripping Calcium Metal in Potassium Hexafluorophosphate Electrolyte toward a Stable Hybrid Solid Electrolyte Interphase. ACS Appl Energy Mater. 2023 Mar 30;6(7):3924-3932. doi: 10.1021/acsaem.3c00098. PMID: 37064409; PMCID: PMC10091900.
[3]Li, Jingru et al. “Potassium Hexafluorophosphate Additive Enables Stable Lithium-Sulfur Batteries.” ACS applied materials & interfaces vol. 12,50 (2020): 56017-56026. doi:10.1021/acsami.0c17406
You may like
See also
Lastest Price from Potassium hexafluorophosphate manufacturers
US $0.00-0.00/kg2026-01-21
- CAS:
- 17084-13-8
- Min. Order:
- 1kg
- Purity:
- 99%
- Supply Ability:
- 1ton

US $70.00/kg2025-04-15
- CAS:
- 17084-13-8
- Min. Order:
- 1kg
- Purity:
- 99
- Supply Ability:
- 5000