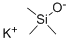Potassium Trimethylsilanolate: Reagent for Ester Cleavage & Cross-Coupling
Potassium trimethylsilanolate is a potent nucleophilic reagent that facilitates the dealkylation of esters under mild, often anhydrous conditions. So this substance is particularly valuable for substrates that are sensitive to acidic or strongly basic aqueous environments. The reaction typically proceeds in aprotic solvents like tetrahydrofuran (THF) at room temperature, offering high to quantitative yields of the corresponding carboxylic acid. For researchers, scientists, and drug development professionals, the efficient and selective cleavage of esters is a critical transformation in organic synthesis. While classical methods like saponification are widely used, the demand for milder conditions and greater functional group tolerance has led to the exploration of alternative reagents. Potassium trimethylsilanolate has emerged as a powerful and versatile tool for the conversion of esters to carboxylic acids. This guide provides a comprehensive comparison of KOSiMe3 with other common ester cleavage methods.

Potassium Trimethylsilanolate-Promoted, Suzuki-Miyaura Cross-Coupling Reaction
The development of transition-metal catalyzed, cross-coupling reactions has fundamentally changed the way chemists construct C–C bonds. Given the importance of the Suzuki-Miyaura reaction, the mechanism by which it proceeds is of considerable interest, as mechanistic understanding allows chemists to better optimize the process. At the most basic level, the Suzuki-Miyaura reaction involves three steps – oxidative addition, transmetalation, and reductive elimination. Inspired by prior work in the Denmark laboratory on cross-coupling reactions of organosilicon reagents, potassium trimethylsilanolate (TMSOK) was identified as a base to promote the homogeneous, anhydrous Suzuki-Miyaura cross-coupling of boronic esters. It was demonstrated that the reaction time for several example reactions could be shortened from days to hours, without changing the ligand or precatalyst, simply by employing an appropriate boronic ester as the nucleophile, potassium trimethylsilanolate as the base, and anhydrous, ethereal solvent. The homogeneous, anhydrous reaction conditions have also been adapted to the cross-coupling of heteroaryl nucleophiles with heteroaryl electrophiles, wherein the use of aqueous conditions can facilitate protodeboronation. As mentioned previously, the Suzuki-Miyaura reaction is frequently carried out using boronate complexes that are poorly soluble in organic media. In contrast, potassium trimethylsilanolate -ligated boronates are highly soluble in THF. It was of interest to uncover what effect a soluble boronate has on the reaction mechanism, particularly with regard to the formation of a Pd-O-B intermediate.[1]
In view of the longstanding assumption that the oxo-palladium (path B) mechanism is highly preferred for access to the pre-transmetalation intermediate, some comment on the demonstration that the boronate pathway (path A) dominates in this process is warranted. In support of the oxo-palladium pathway, Hartwig and Carrow demonstrated that transmetalation from potassium triolboronates is very slow at −40 °C. In contrast, the rate of transmetalation from potassium trimethylsilanolate-ligated boronic esters is rapid at −40 °C. We hypothesize that in this system, the viability of boronate transmetalation pathway (path A) from TMSOK-complexes of boronic esters is related to the solubility of the potassium boronate and the insolubility of the inorganic salt byproduct, KBr. The solubilized potassium cation could bind as a Lewis acid to the bromide in complexes, activating the arylpalladium bromide complex to transmetalation. A possible explanation for a lack of reactivity through the oxo-palladium pathway observed herein is that the high affinity of TMSOK for boron and low affinity for palladium prevents the appropriate speciation to enable that pathway. The mechanism of the potassium trimethylsilanolate-promoted, anhydrous Suzuki-Miyaura cross-coupling reaction has been elucidated through kinetic analysis and the study of reactivity of isolated reaction intermediates. Stoichiometric studies show that in contrast to prior results, transmetalation through the boronate pathway (path A) can proceed quickly under appropriate reaction conditions.
Validation of the Existence of Tetrameric Species of Potassium Trimethylsilanolate
A considerable number of recent studies deal with the synthesis and structural characterization of different metallic alkylsilanolates. These compounds have increasingly attracted the attention of several disciplines, such as material science and catalysis, due to their potential applications in the preparation of inorganic materials with improved physical properties. Although extensively studied experimentally (mainly by NMR and X-ray diffraction techniques), theoretical investigations on these structures are rare. In recent years, however, there has been a growing interest in combined theoretical and experimental studies that deal with the bonding properties and molecular structures of compounds containing highly polar bonds of the type M−X (where M = metal and X = C, O, ...). The system studied in the present work, potassium trimethylsilanolate, belongs to this family of compounds. The crystal and molecular structures of potassium trimethylsilanolate have been studied previously by X-ray diffraction (XRD) and mass spectrometry. Weiss et al. were first to solve the crystal structure of this compound, from powder XRD data, in the cubic space group P4̄3m (R = 7.9%). In the Weiss structure, potassium trimethylsilanolate forms tetrameric units where the oxygen and potassium atoms are arranged in a slightly distorted cube. The authors also suggested the existence of these tetrameric units in the gas phase from mass spectrometry experiments.[2]
The existence and stability of the tetrameric unit of potassium trimethylsilanolate in the gas phase has been validated theoretically using a simplified ionic cluster model, a result that could encourage the experimentalists to try the isolation of these ionic clusters of metallic silanolates. Since the B3LYP/6-31+G* optimized structural parameters for the model reproduced the experimental XRD ones reported in Pauer's study,5 with the exception of the SiO bond length (whose calculated value was in closer agreement to that in ref 4), the validity of the model has been demonstrated. The optimized anti conformation in the gas phase of the trimethylsilanolate and silyl groups in the cluster model is in disagreement with the experimental observations in the solid phase. The optimization at the same level of a simpler cluster, stoichiometry K4(OSiH3)4, confirmed the preferred anti orientation of the −SiH3 groups since four negative frequencies (corresponding to the deformation vibrations of the silyl group) were obtained when the eclipsed conformation was imposed. The comparative analyses of the optimized geometries calculated for the isolated anion, a monomer, and the simplified ionic cluster with the experimental structure of the potassium trimethylsilanolate salt revealed the importance of considering the counterion implicitly in the models. NBO calculations of the electronic structure corroborated point 4 as a certain charge from the lone pairs of the oxygen and the SiO bonding orbitals was localized in the potassium atoms.
Cross-Coupling Promoted by Potassium Trimethylsilanolate
Conjugated acetylenes play a major role in synthetic, pharmaceutical, and polymer chemistry. One of the most widely used methods for the construction of conjugated acetylenes is the Sonogashira reaction. The development of alternative methods for activation is desirable because fluoride reagents carry the dual disadvantages of high reagent cost and poor compatibility with silicon-based protecting groups. Recent reports from these and other laboratories have described methods for the palladium-catalyzed cross-coupling of silanols under fluoride-free conditions. These studies have shown that potassium trimethylsilanolate (KOTMS)15b can promote cross-coupling of alkenylsilanols (at rates comparable to TBAF) under conditions that are compatible with silicon-based protecting groups. The palladium-catalyzed cross-coupling of aliphatic alkynylsilanols with aryl iodides has been demonstrated with potassium trimethylsilanolate as the coupling promoter and copper(I) iodide as a cocatalyst. The cross-coupling proceeds at room temperature in good to excellent yield with a range of aryl iodides. A comparison of the reactivity of alkynylsilanols, trimethylsilylalkynes, and terminal alkynes under fluoride and fluoride-free conditions was performed to elucidate the role of silicon in the Sonogashira reaction.[3]
References
[1]Delaney CP, Marron DP, Shved AS, Zare RN, Waymouth RM, Denmark SE. Potassium Trimethylsilanolate-Promoted, Anhydrous Suzuki-Miyaura Cross-Coupling Reaction Proceeds via the "Boronate Mechanism": Evidence for the Alternative Fork in the Trail. J Am Chem Soc. 2022 Mar 16;144(10):4345-4364. doi: 10.1021/jacs.1c08283. Epub 2022 Mar 1. PMID: 35230833; PMCID: PMC8930609.
[2]Montejo, Manuel et al. “Validation of the existence of tetrameric species of potassium trimethylsilanolate in the gas phase with a theoretical cluster model: role of the counterion as charge localizer in the structure.” The journal of physical chemistry. A vol. 111,13 (2007): 2629-33. doi:10.1021/jp0686240
[3]Denmark, Scott E, and Steven A Tymonko. “Cross-coupling of alkynylsilanols with aryl halides promoted by potassium trimethylsilanolate.” The Journal of organic chemistry vol. 68,23 (2003): 9151-4. doi:10.1021/jo0351771
You may like
Related articles And Qustion
See also
Lastest Price from Potassium trimethylsilanolate manufacturers

US $0.00-0.00/KG2025-04-15
- CAS:
- 10519-96-7
- Min. Order:
- 1KG
- Purity:
- 99%
- Supply Ability:
- 500000kg

US $20.00-1.00/kg2025-03-07
- CAS:
- 10519-96-7
- Min. Order:
- 1kg
- Purity:
- 0.99
- Supply Ability:
- 30 tons