Preparation and application research of 1,3-Bis(aminomethyl)benzene
Introduction
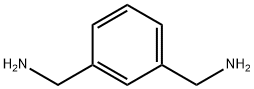
1,3-Bis(aminomethyl)benzene (also known as m-xylylenediamine or m-Benzodimethylamine;Figure 1) is an important fine chemical intermediate. 1,3-Bis(aminomethyl)benzene is a colorless, transparent liquid with an almond-like odor at room temperature. It is readily soluble in water, alcohols, benzene, and most other organic solvents. 1,3-Bis(aminomethyl)benzene was found to have great potential intissue clearing. 1,3-Bis(aminomethyl)benzene is a colorless and water miscible liquidwith good fluidity and has two NH2 groups similar to urea, it is expected to have great hyperhydration ability and is easy to penetrate into tissue. Additionally, 1,3-bis(aminomethyl)benzene has a high RI up to 1.57, which is critical for RI matching in tissue clearing procedure. With excellent physical and chemical properties, 1,3-bis(aminomethyl)benzene has a wide range of applications.

It plays an important role in many fields, including dyes, epoxy resin curing agents, cement accelerators, petroleum additives, and the manufacture of pharmaceuticals and pesticides. Furthermore, owing to the presence of two primary amine groups in its molecule, 1,3-bis(aminomethyl)benzene can participate in a variety of chemical reactions. It is also widely used as rubber crosslinking agents, fiber stabilizers, and surfactants, making it one of the important fine chemicals urgently needed for development in China.
Preparation of 1,3-Bis(aminomethyl)benzene
Report 1: Lv et al. had introduced a comprehensive organic chemistry experiment: the reductive amination of isophthalaldehyde with silane to synthesize 1,3-bis(aminomethyl)benzene. In this reaction, tert-butyl carbamate (BocNH2 ) serves as the nitrogen source, and triethylsilane (Et3SiH) acts as the reducing agent, with trifluoroacetic acid as the catalyst. Isophthalaldehyde undergoes reductive amination to produce Boc-protected 1,3-bis(aminomethyl)benzene, which is subsequently deprotected to yield 1,3-bis(aminomethyl)benzene hydrochloride. The final product, 1,3-bis(aminomethyl)benzene, is obtained by treating the hydrochloride salt with sodium hydroxide. The experimental procedure includes techniques such as extraction, vacuum distillation, drying, recrystallization, and melting point determination, along with analytical methods like thin-layer chromatography (TLC), infrared (IR) spectroscopy, and nuclear magnetic resonance (NMR) spectroscopy. This synthetic route is well-suited for laboratory synthesis, providing high yields and easy purification of the product.
Report 2: The industrial production of 1,3-bis(aminomethyl)benzene mainly uses m-xylene as raw material, obtains m-phenyldinitrile through ammoxidation, and then hydrogenates to produce 1,3-bis(aminomethyl)benzene. During the processes, the hydrogenation of m-phenyldinitrile needs to be performed under high temperature and pressure with high energy consumption. Also, complicated processes of inhibitors separation and products purification in the later stage cause low product yield. Therefore, it is of great significance to develop efficient catalysts for hydrogenation of m-phenyldinitrile. In this report, metal nickel was used as active catalyst to construct a highly-active catalytic system by loading the catalyst on some support and introducing metal cocatalyst. The hydrogenation of m-phenyldinitrile with high conversion and high selectivity for 1,3-bis(aminomethyl)benzene was realized in the absence of sodium hydroxide, ammonia water and high-pressure liquid ammonia.
Nickel based catalysts were supported on different carriers by deposition and precipitation method. The high-activity catalyst of 10 wt% Ni-γ-Al2O3 was prepared by optimizing the loading amount of the catalysts. The γ-Al2O3 carrier has a high specific surface area, which enables nickel to be highly dispersed. The uniform loading of nickel nanoparticles can greatly accelerate the hydrogen activation rate. By optimizing the reaction conditions, m-phenyldinitrile can be converted 100% and higher than 99% of selectivity for dimethylamine can be obtained at 140℃ with 3 MPa H2 after 3h reaction.[2]
Application research
1,3-Bis(aminomethyl)benzene-based Aqueous Clearing System preparation
Tissue optical clearing techniques have provided important tools for large-volume imaging. Aqueous-based clearing methods are known for good fluorescence preservation and scalable size maintenance, but are limited by long incubation time, insufficient clearing performance, or requirements for specialized devices. Additionally, few clearing methods are compatible with widely used lipophilic dyes while maintaining high clearing performance. Here, to address these issues, 1,3-bis(aminomethyl)benzene is firstly introduced into tissue clearing and used to develop a rapid, highly efficient aqueous clearing method with robust lipophilic dyes compatibility, termed 1,3-bis(aminomethyl)benzene-based Aqueous Clearing System (MACS). MACS can render whole adult brains highly transparent within 2.5 days and is also applicable for other intact organs. Meanwhile, MACS possesses ideal compatibility with multiple probes, especially for lipophilic dyes. MACS achieves 3D imaging of the intact neural structures labeled by various techniques. Combining MACS with DiI labeling, MACS allows reconstruction of the detailed vascular structures of various organs and generates 3D pathology of glomeruli tufts in healthy and diabetic kidneys. Therefore, MACS provides a useful method for 3D mapping of intact tissues and is expected to facilitate morphological, physiological, and pathological studies of various organs.[3]
1,3-Bis(aminomethyl)benzene as the nitrogen source
Preparation of water-dispersible carbon dots from inexpensive natural carbon precursors and its application for purposes such as sensing, bio-imaging and patterning agents is showing growing interest in recent years. Bhatt et al. have reported the preparation of nitrogen-doped carbon dots (N-CDs) using prickly pear as the carbon source and 1,3-bis(aminomethyl)benzene as the nitrogen source using a one-step microwave-assisted synthetic process. The N-CDs prepared were characterized on the basis of elemental analysis, XPS, powder-XRD, FT-IR, Raman, TEM, UV-vis and fluorescence spectroscopy. Doping of nitrogen in the N-CDs made them highly fluorescent and the study on their ion-recognition property revealed that they detect highly toxic Cr(VI) with high selectivity and sensitivity (LOD, 0.04 μM) and without interference from the other ions used in this study. By immobilizing these N-CDs onto filter paper, sensor strips were prepared for on-site monitoring/field applications and they were successfully used for the detection of Cr(VI) in water. Detailed spectral analysis revealed that the mechanism of Cr(VI) sensing involved a phenomenon called the "inner filter effect" and analysis of the fluorescence lifetime data suggested the "static quenching" of fluorescence intensity. These N-CDs were used to prepare fluorescent carbon ink and were successfully used as patterning agents.[4]
References
[1] Lv LY, et al.,Reductive Amination Reaction: Preparation of m-Xylylenediamine from Isophthalaldehyde as an Example[J].University Chemistry,2024,39(12):258-264.
[2] Sun GF.Preparation of m-Benzodimethylamine by catalytic hydrogenation of m-phenyldinitrile[D].Northwest Normal University ,2023.DOI:10.27410/d.cnki.gxbfu.2023.002424.
[3] Zhu J, Yu T, Li Y, et al. MACS: Rapid Aqueous Clearing System for 3D Mapping of Intact Organs. Adv Sci (Weinh). 2020;7(8):1903185. Published 2020 Feb 25. doi:10.1002/advs.201903185
[4] Bhatt S, Vyas G, Paul P. Microwave-assisted synthesis of nitrogen-doped carbon dots using prickly pear as the carbon source and its application as a highly selective sensor for Cr(VI) and as a patterning agent. Anal Methods. 2022;14(3):269-277. Published 2022 Jan 20. doi:10.1039/d1ay01274f
You may like
Related articles And Qustion
Lastest Price from 1,3-Bis(aminomethyl)benzene manufacturers

US $10.00/KG2025-04-21
- CAS:
- 1477-55-0
- Min. Order:
- 1KG
- Purity:
- 99%
- Supply Ability:
- 100 mt

US $79.00-38.00/kg2025-04-21
- CAS:
- 1477-55-0
- Min. Order:
- 1kg
- Purity:
- 99%
- Supply Ability:
- 20ton