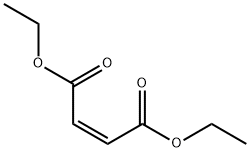
Preparation and Biological Activity of Diethyl maleate
Diethyl maleate is a colorless transparent liquid under ambient temperature and pressure, miscible with a wide range of organic solvents, partially soluble in benzene and chloroform, and exhibits a solubility of 1.4% in water at 30°C, while water shows a solubility of 1.9% in diethyl maleate. It forms an azeotropic mixture with 88.2% water, with an azeotropic point of 99.65°C. In the field of chemical production, Diethyl maleate is primarily used as an intermediate in pesticide manufacturing—specifically for the synthesis of the organophosphorus pesticide malathion—and is also employed in the production of fragrances.

Figure1: Picture of Diethyl maleate
Preparation Methods
Diethyl maleate can be prepared by the esterification of maleic anhydride with ethanol in the presence of sulfuric acid, or through exchange conversion using a cationic exchange resin as the catalyst. The industrial-grade product typically contains ≥98% diethyl maleate, with consumption rates of approximately 585 kg of maleic anhydride (95% purity) and 604 kg of ethanol (95% purity) per ton of product. Two main processes are employed in industrial production: atmospheric-pressure esterification with benzene and reduced-pressure benzene-free esterification. [1]
Atmospheric-pressure Esterification with Benzene
A measured amount of benzene and ethanol is charged into an esterification reactor, followed by the addition of maleic anhydride. Concentrated sulfuric acid is then added dropwise under stirring, and the mixture is heated via a jacket steam system to maintain the reaction temperature around 75°C. The water generated during esterification is removed by ternary azeotropic distillation along with benzene and ethanol, while the upper benzene–ethanol layer is refluxed back into the reactor. After approximately 13–14 hours, when the distillation column head temperature reaches 68.2°C and the water level in the separator no longer increases, it indicates complete removal of water and completion of esterification. Reflux is then stopped, and distillation is continued at 95–100°C to recover benzene and ethanol. After cooling to about 50°C, the crude mixture is neutralized with a 5% sodium carbonate solution, washed with water, and subjected to vacuum treatment to remove residual benzene and ethanol, yielding the final product Diethyl maleate.
Reduced-pressure Benzene-free Esterification
In this method, maleic anhydride and ethanol undergo esterification under sulfuric acid catalysis while applying controlled vacuum and temperature. Ethanol and the generated water are vaporized and carried out of the system, followed by separation in a fractionation column; the recovered ethanol is then returned to the reactor to drive the reaction toward completion. This approach shortens the reaction cycle, improves yield and product quality, and provides a better operational environment, making it the preferred method for most domestic manufacturers. Alternatively, Diethyl maleate can also be synthesized via exchange conversion catalyzed by a cationic exchange resin. Purification Method: The crude Diethyl maleate product is washed with a dilute potassium carbonate solution, dried over anhydrous potassium carbonate or sodium sulfate, and then subjected to vacuum distillation for further purification.
Biological Activity
Diethyl maleate (DEM) is an orally available, effective glutathione (GSH) depletor that crosses the blood-brain barrier. Diethyl maleate covalently binds irreversibly to GSH via glutathione S-transferase with an in vitro IC50 of 0.1-0.5 mM. Diethyl maleate selectively depletes GSH in liver, lung, and brain tissues, exacerbating oxidative stress and enhancing hyperbaric oxygen toxicity. Diethyl maleate promotes precursor amino acid uptake and in turn promotes GSH synthesis by upregulating the activity of the cystine-glutamate transporter XO-. Diethyl maleate can be used to study redox homeostasis and GSH protection mechanisms in oxidative stress-related diseases such as hyperbaric oxygen injury and metabolic diseases.
Application
Research has reported the application of diethyl maleate as a fungicide synergist in plant disease control. Specifically, this fungicide synergist is used in combination with substituted aniline fungicides, methoxyacrylate fungicides, triazole fungicides, and substituted benzene fungicides for disease management. Diethyl maleate exhibits a synergistic effect when mixed with these fungicides, enhancing their toxicity, increasing the sensitivity of resistant strains to the fungicides, and improving control efficacy against plant pathogens that have developed resistance in both laboratory and field conditions. Furthermore, diethyl maleate can effectively reduce the required dosage of fungicides, providing a novel approach for the development and application of new formulations of existing fungicides. [2]
Reference
[1] You P Q, Li W Y, Wen R M, et al. Sodium hydrogen sulfate catalyzed synthesis of diethyl maleate[J]. Industrial Catalysis, 2004.
[2] Liu P F, et al. Application of diethyl maleate as fungicide synergist in plant disease control: CN202110836074.9[P].
You may like
See also
Lastest Price from Diethyl maleate manufacturers

US $0.00/kg2025-07-09
- CAS:
- 141-05-9
- Min. Order:
- 1kg
- Purity:
- 98%min
- Supply Ability:
- 20 tons

US $0.00/KG2025-04-15
- CAS:
- 141-05-9
- Min. Order:
- 1KG
- Purity:
- 99%
- Supply Ability:
- 500000kg