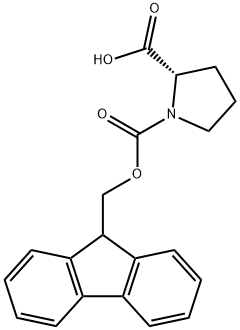
Preparation Method and Biological Applications of Fmoc-Pro-OH
Fmoc-Pro-OH is a chiral amino acid compound that appears as a white to off-white solid powder at room temperature and pressure, exhibiting good chemical stability. It is insoluble in water but soluble in alcohol-based organic solvents. Fmoc-Pro-OH can be prepared from L-proline and 9-fluorenylmethyl chloroformate or 9-fluorenylmethyl succinimidyl carbonate via a one-step condensation reaction. This substance is one of the main components of collagen in connective tissue, and its content determines the amount of muscle collagen, which is closely related to tenderness.
Preparation Method
Method 1
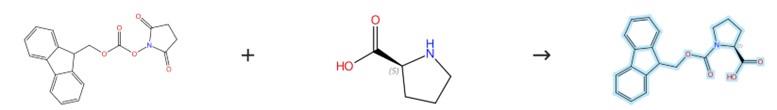
Figure1: Preparation Method of Fmoc-Pro-OH
L-Proline (1 g) is dissolved in a mixture of 1,4-dioxane (4 mL) and water (15 mL), and the resulting solution is cooled to 0 °C before potassium carbonate (3.24 g, 23 mmol) is added, followed by the portionwise addition of (9H-fluoren-9-yl)methyl chloroformate (2.3 g), after which the reaction mixture is stirred at room temperature overnight. Upon the addition of water (10 mL), the mixture is extracted with diethyl ether (2 × 20 mL), and the aqueous phase is subsequently acidified with 1 M aqueous hydrochloric acid to pH 2–3 and then extracted with dichloromethane (3 × 50 mL). The combined organic extracts are finally dried over anhydrous sodium sulfate, filtered, and concentrated to dryness to give the product Fmoc-Pro-OH. [1]
Method 2
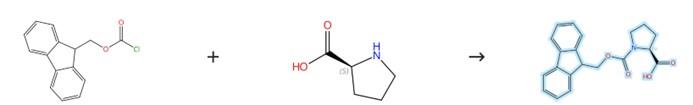
Figure2: Preparation Method of Fmoc-Pro-OH
L‑Proline (1.5 g) is dissolved in 30 mL of an aqueous sodium carbonate solution (10% by mass) under stirring, and this mixture is then combined with a solution of 4.5 g of N‑(9‑fluorenylmethoxycarbonyloxy)succinimide in tetrahydrofuran (30 mL). After the reaction has proceeded for 48 hours, water (40 mL) is added, and the resulting mixture is extracted with diethyl ether (5 × 30 mL); the aqueous phase is subsequently adjusted to pH 2–3 with aqueous hydrochloric acid and extracted with ethyl acetate (5 × 30 mL). The combined organic layers containing the desired product are then dried over anhydrous sodium sulfate overnight, followed by removal of the ethyl acetate under reduced pressure by evaporation to give the product Fmoc-Pro-OH. [2]
Preparation of Tripeptide Cellulose Ester
Using microcrystalline cellulose as the raw material, a tripeptide cellulose ester (TPC) with a modified amino terminus was synthesized via acyl chloride formation, esterification, peptide chain elongation, and amino group modification, employing 9-fluorenylmethoxycarbonyl-L-proline (Fmoc-Pro-OH), 9-fluorenylmethoxycarbonyl-L-valyl-L-phenylalanine, and 3,5-dinitrobenzoic acid as derivatization reagents. Fmoc-Pro-OH was essential in the peptide chain elongation step, ensuring stereochemical integrity during synthesis. The optimal esterification conditions were a cellulose concentration of 20 g/L in LiCl/DMAc at 100 °C, an acyl chloride to hydroxyl molar ratio of 3:1, and a reaction time of 20 h, yielding a product with a degree of substitution of 2.15. Characterization confirmed successful modification of the peptide terminus with 3,5-dinitrobenzoic acid. Depending on the precipitation solvent, the product exhibited different hydrogen bonding patterns and functionalities. Fmoc-Pro-OH also contributed to the chiral recognition properties of the final material. Application studies showed that TPC precipitated with methanol served as an effective enteric coating with strong light-blocking properties and rapid disintegration, while TPC precipitated with water, when used in chiral coated columns, combined the advantages of brush-type and cellulose stationary phases, offering superior enantioseparation performance, excellent chemical resistance, and broader applicability, especially with tetrahydrofuran-containing mobile phases. [3]
Biological Applications
Fmoc-Pro-OH can be used as a chiral synthesis intermediate and a basic chemical raw material for pharmaceutical synthesis. For example, it has been reported in the literature to be used in the synthesis of insect neuropeptide analogs of the AKH family. Fmoc-Pro-OH can also be used in the synthesis of insect pyrokinin neuropeptides having activity against insects, and their use as insect control and plant protection agents.
Reference
[1] Veenstra, Marieke J.; et al, Organocatalyst based cross-catalytic system, Chemical Communications (Cambridge, United Kingdom) 2022, 58, 13895-13898.
[2] Guo, Guozhang; et al, Polymerization of L-proline functionalized styrene and its catalytic performance as a supported organocatalyst for direct enantioselective aldol reaction, Tetrahedron: Asymmetry 2016, 27, 740-746.
[3] Li Yang, Zhu Jinke, Li Lianjie, et al. Preparation and Application of Tripeptide Cellulose Ester[J]. Chemistry and Industry of Forest Products, 2016, 36: 8.
You may like
Lastest Price from Fmoc-Pro-OH manufacturers
US $0.00/kg2025-09-22
- CAS:
- 71989-31-6
- Min. Order:
- 1kg
- Purity:
- 99%
- Supply Ability:
- 1000kg

US $100.00-2500.00/KG2025-06-17
- CAS:
- 71989-31-6
- Min. Order:
- 1KG
- Purity:
- 99%
- Supply Ability:
- 10MT per month