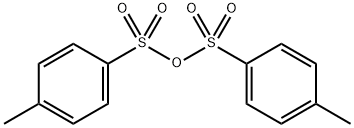
Preparation Method and Chemical Properties of 4-Methylbenzenesulfonic Anhydride
4-Methylbenzenesulfonic Anhydride is a sulfonic anhydride compound that appears as a yellow to light gray solid powder at room temperature and under atmospheric pressure. It is highly hygroscopic and prone to deliquescence, and it readily undergoes hydrolysis and decomposition upon exposure to water. 4-Methylbenzenesulfonic Anhydride can be prepared via the acylation of p-toluenesulfonyl chloride with p-toluenesulfonic acid. This compound is primarily used as an acylating agent, especially for the acylation reactions of organic alcohols or amine compounds, and it has good applications in the field of basic research in organic synthesis methodology.
Preparation Method
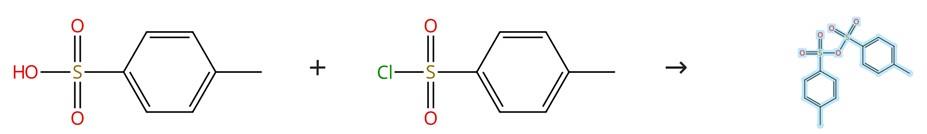
Figure 1: Synthesis of 4-Methylbenzenesulfonic Anhydride
Disperse a mixture consisting of p-toluenesulfonyl chloride (16 g) and p-toluenesulfonic acid monohydrate (4 g) in 100 mL of methylene chloride (dichloromethane), obtained from Scharlab, Barcelona, Spain. Allow the resulting reaction mixture to stir continuously at room temperature overnight. Following the stirring period, filter the suspension in order to remove any unreacted p-toluenesulfonyl chloride that remains in the mixture. Collect the filtrate solution and concentrate it by evaporating the solvent under reduced pressure. Subsequently, dry the obtained solid under vacuum overnight, which yields the final product: 4-Methylbenzenesulfonic Anhydride. [1]
Acylation Reaction
4-Methylbenzenesulfonic Anhydride possesses significant electrophilicity and can undergo acylation reactions with common alcohols and organic amines under basic conditions. It is widely used for the protection of alcohol and organic amine compounds.
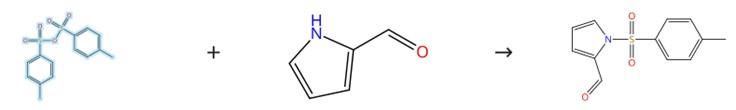
Figure 2: Acylation Reaction of 4-Methylbenzenesulfonic Anhydride
To a flame-dried 50 mL round-bottom flask equipped with a magnetic stir bar is added 1H-pyrrole-2-carbaldehyde (1.142 g, 12.00 mmol, 1 equivalent), 4-(dimethylamino)pyridine (146.6 mg, 1.2 mmol, 0.1 equivalent), and 30 mL of anhydrous dichloromethane (DCM), after which 4-Methylbenzenesulfonic Anhydride (1.05 equivalents relative to the aldehyde) is introduced into the resulting solution; the flask is then securely sealed with a septum and the reaction mixture is allowed to stir continuously at room temperature overnight. Following the overnight stirring period, the reaction is quenched by the addition of 20 mL of brine, and the aqueous layer is further extracted with two separate 10 mL portions of DCM. The combined organic layers are subsequently washed with a single 40 mL portion of brine, and the resulting organic mixture is dried over anhydrous sodium sulfate. Finally, the residue obtained after drying is purified by column chromatography using a gradient elution system that increases from 0% to 40% ethyl acetate in hexanes. [2]
Enolization reaction
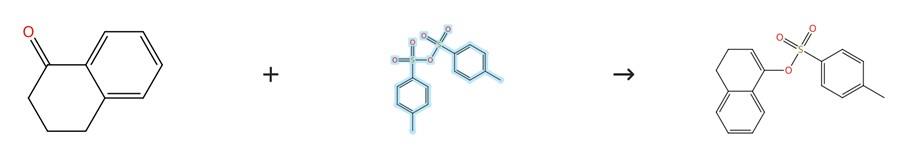
Figure 3: 4-Methylbenzenesulfonic Anhydride-Promoted Enolization Reaction
Add hexamethyldisilazane (HMDS, 1.3 equivalents) to tetrahydrofuran (THF) at a concentration of 2.3 mmol of HMDS per milliliter of THF, and then cool the resulting solution to −78 °C. To this chilled solution containing HMDS, add a solution of n-butyllithium (1.2 equivalents, provided as a solution in hexanes) dropwise over a period of 5 minutes, and stir the mixture for 30 minutes while maintaining the temperature at −78 °C. Next, add a solution of α-tetralone (1.0 equivalent, prepared at a concentration of 10.0 mmol of ketone per milliliter of THF) dropwise to the solution containing the freshly prepared lithium hexamethyldisilazide (LiHMDS) over 5–10 minutes, and continue stirring the flask for an additional 1 to 1.5 hours at −78 °C. After this allotted time has elapsed, add a solution of 4-Methylbenzenesulfonic Anhydride (Ts₂O, 1.4 equivalents, dissolved in THF at a concentration of 0.70 mmol of 4-Methylbenzenesulfonic Anhydride per milliliter of THF) to the solution containing the enolate, and then allow the reaction mixture to gradually warm to room temperature over the course of the addition. Once at room temperature, quench the reaction by adding saturated aqueous ammonium chloride (NH₄Cl), and leave the reaction to stir overnight. Following the overnight stirring, extract the reaction mixture three times with ethyl acetate, remove the combined solvent under reduced pressure, add hexanes to the residue, dry the resulting mixture over anhydrous magnesium sulfate (MgSO₄), and sonicate the suspension. Finally, purify the product by column chromatography. [3]
Reference
[1] Torres-Ortega, Pablo Vicente; et al, Encapsulation of MSCs and GDNF in an Injectable Nanoreinforced Supramolecular Hydrogel for Brain Tissue Engineering, Biomacromolecules 2022, 23, 4629-4644.
[2] Akkawi, Nicholas R.; et al, Photochemically enabled total syntheses of stemoamide alkaloids, Journal of the American Chemical Society 2025, 147, 15482-15489.
[3] Graham, Joshua M.; et al, Ni-catalyzed reductive cyanation of alkenyl tosylates and triflates, Chemical Communications 2025, 61, 893-896.
You may like
Lastest Price from P-Toluenesulfonic anhydride manufacturers
US $0.00-0.00/kg2025-11-20
- CAS:
- 4124-41-8
- Min. Order:
- 1kg
- Purity:
- 98%
- Supply Ability:
- 100tons

US $10.00/kg2025-04-21
- CAS:
- 4124-41-8
- Min. Order:
- 1kg
- Purity:
- 99%
- Supply Ability:
- 20 ton