Preparation Method and Nucleophilic substitution reaction of 1,5-Dibromopentane
1,5-Dibromopentane is an alkyl bromide compound. At room temperature and pressure, it appears as a clear, colorless to yellow-brown liquid with a relatively high density. It is insoluble in water but miscible with most organic solvents. In the field of chemical synthesis, 1,5-dibromopentane is commonly used as an organic synthesis intermediate. It can be attacked twice by nucleophiles and is useful in the preparation of cyclopentane derivatives.
Preparation Method
Method 1
Prepared by the ring-opening bromination of tetrahydropyran. In a 500 mL flask, sequentially add 250 g of 48% hydrobromic acid, 75 g of concentrated sulfuric acid, and 21.5 g of redistilled tetrahydropyran (86.5–87.5 °C fraction). Attach a reflux condenser and gently heat the brown mixture under mild reflux for 3 hours. After cooling to room temperature, separate the lower dibromide layer and wash it once with saturated sodium carbonate solution and once with water. Dry over anhydrous calcium chloride. Distill under reduced pressure, collecting the fraction at 104–106 °C (2.53 kPa) as the final product 1,5-Dibromopentane. The yield is 46–47 g, corresponding to an 80–82% yield.
Method 2
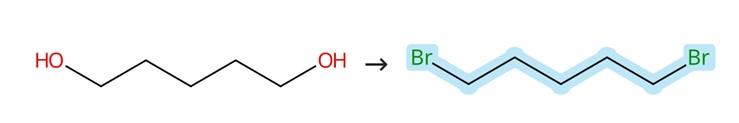
Figure1: Synthesis of 1,5-Dibromopentane
Under an ice bath, 168 mL of 48% HBr, 41 mL of 98% H₂SO₄, and 36 mL of 1,5-pentanediol were slowly and successively added into a 500 mL round-bottomed flask, after which an additional 32.5 mL of 98% H₂SO₄ was slowly introduced to the reaction mixture. The resulting mixture was then heated under reflux at 120 °C for a period of 3 hours to ensure complete reaction. Following the reflux step, the organic phase was carefully separated from the aqueous layer, and subsequently washed sequentially with water, 10% HCl, water again, and finally with saturated sodium bicarbonate solution to remove any residual acidic impurities. The resulting organic mixture was then subjected to distillation, yielding the final product, 1,5-Dibromopentane.[1]
Synthesis of Wittig reagent
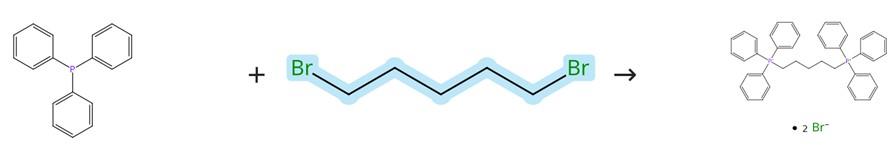
Figure2: Synthesis of Wittig reagent
Synthesis of Wittig reagent Triphenylphosphine (57.7 g, 220 mmol) was first dissolved in 300 mL of acetonitrile, and then 1,5-Dibromopentane (23.0 g, 5.5 mL, 100 mmol) was added to the resulting solution. The mixture was heated under reflux for a period of 5 days to ensure complete reaction, after which the solvent was evaporated to obtain the crude product. This crude product was subsequently dissolved in 100 mL of hot dichloromethane (DCM), followed by the addition of 250 mL of ethyl acetate (EtOAc), which led to the formation of a precipitate that was collected by filtration. The obtained precipitate was thoroughly washed with diethyl ether and finally dried under reduced pressure to yield the purified product. [2]
Nucleophilic substitution reaction
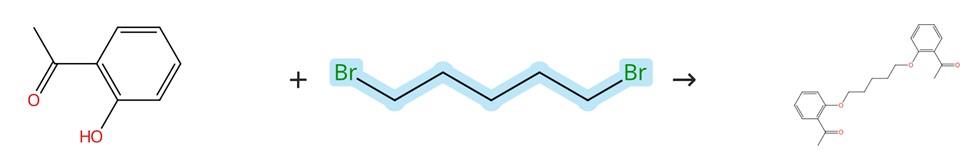
Figure3: Nucleophilic substitution reaction of 1,5-Dibromopentane
To carry out the Williamson ether synthesis, two equivalents (20 mmol) of 2-hydroxyacetophenone, one equivalent (10 mmol) of the corresponding 1,5-Dibromopentane, and five equivalents of K₂CO₃ were refluxed in ethanol for 8 hours, and the progress of the reaction was continuously monitored by FTIR spectroscopy until completion. Once the reaction was complete, the resulting solution was allowed to cool, and cold distilled water was added to precipitate a solid product. The mixture was then filtered, and the collected solid was thoroughly washed several times with distilled water and cold ethanol to remove any impurities. After washing, the residue was dried, followed by recrystallization in ethanol, yielding the final purified product: 1,1'-((pentane-1,5-diylbis(oxy))bis(2,1-phenylene))bis(ethan-1-one). [3]
Cyclization reaction
A mixture containing mesitylamine (19.5 g, 144 mmol), 1,5-Dibromopentane (27.6 g, 120 mmol), K₂CO₃ (36.5 g, 264 mmol), and NaI (39.6 g, 264 mmol) in CH₃CN (400 mL) was stirred under reflux conditions for 48 hours to ensure complete reaction. After the mixture had cooled to room temperature, diethyl ether (Et₂O) was added, and the resulting precipitate was removed by filtration. All volatile components in the filtrate were then evaporated under reduced pressure, and the obtained residue was subsequently purified by flash silica gel column chromatography using hexane as the eluent. [4]
Reference
[1] Wang, Tsung-Shing Andrew ; et al, Selectively Targeting and Differentiating Vancomycin-Resistant Staphylococcus aureus via Dual Synthetic Fluorescent Probes, ACS Infectious Diseases 2021, 7, 2584-2590.
[2] Albitz, Krisztian ; et al, Unlocking Oxetane Potential: Modular Synthetic Platform for the Concise Synthesis of Acyclic Oligo-Isoprenoids and Terpenoids, Angewandte Chemie, International Edition 2025, 64, e202416441.
[3] Aziz, Marlin Y. ; et al, Host-guest chemistry: Novel macrocyclic crown chalcone-ether hybrids; Design, synthesis, characterization, biological evaluations, molecular docking and color removal application, Journal of Molecular Structure 2026, 1354, 144910.
[4] Meng, Xiaodan; et al, Transition metal-free α,β-C(sp3)-H dehydrogenative diazotization of piperidines under mild conditions, Green Chemistry (2026), 28(6), 2694-2699.
You may like
See also
Lastest Price from 1,5-Dibromopentane manufacturers

US $0.00/KG2025-12-28
- CAS:
- 111-24-0
- Min. Order:
- 1KG
- Purity:
- ≥99%
- Supply Ability:
- 50 Tons/Month

US $0.00/KG2025-04-21
- CAS:
- 111-24-0
- Min. Order:
- 1KG
- Purity:
- 98%min
- Supply Ability:
- 30tons/month
