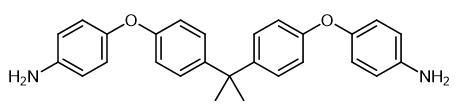
Preparation Method of 2,2-Bis[4-(4-aminophenoxy)phenyl]propane
2,2-Bis[4-(4-aminophenoxy)phenyl]propane is a white solid powder at room temperature and pressure. It exhibits significant basicity and good chemical stability. It is insoluble in water but soluble in alcohol-based organic solvents. 2,2-Bis[4-(4-aminophenoxy)phenyl]propane can be prepared by reacting bisphenol A with p-chloronitrobenzene in DMAc at high temperature, followed by a reduction reaction. This substance is primarily used as a diamine-based curing agent and can be applied in the preparation and development of polyester-based new materials.
Preparation Method
In a 250 mL three-neck flask, BNPP (10 mmol) and 10% Pd/C (0.05 g) were charged in ethanol (150 mL), after which hydrazine monohydrate (10 mL) was added to the stirred mixture at 70 °C over a period of 30 minutes, and following the completion of this addition, the mixture was heated at reflux temperature for 8 hours to facilitate the reduction reaction. 2,2-Bis[4-(4-aminophenoxy)phenyl]propane was obtained after the reaction solution was filtered to remove the Pd/C catalyst, and the crude solid was subsequently recrystallized from ethanol to yield 3.4 g of white crystals (83%), with a melting point of 129 °C as determined by DSC. 2,2-Bis[4-(4-aminophenoxy)phenyl]propane, as the final product of this synthetic procedure, was thus successfully isolated in good yield and high purity.[1]
![Synthesis of 2,2-Bis[4-(4-aminophenoxy)phenyl]propane Article illustration](/NewsImg/2026-03-22/6390981185175611622296370.jpg)
Figure1: Synthesis of 2,2-Bis[4-(4-aminophenoxy)phenyl]propane
Synthetic Procedure
To synthesize the diamine monomer 2,2-Bis[4-(4-aminophenoxy)phenyl]propane, the intermediate dinitro compound (BNPP) was first prepared through an aromatic nucleophilic substitution reaction of p-chloronitrobenzene in the presence of potassium carbonate in DMAc, wherein the electron-withdrawing nitro group of p-chloronitrobenzene activated the chlorine atom for facile displacement. Subsequently, BNPP was reduced using hydrazine hydrate and a Pd/C catalyst in refluxing ethanol to afford the desired product in high yield. The FTIR spectrum of BNPP revealed two characteristic absorption bands at 1510 cm⁻¹ and 1345 cm⁻¹, attributed to the asymmetric and symmetric stretching vibrations of the N=O group, along with an additional band at 1250 cm⁻¹ corresponding to C–N stretching; upon reduction, the characteristic absorptions of the nitro group disappeared, while a pair of N–H stretching bands appeared in the region of 3300–3450 cm⁻¹. 2,2-Bis[4-(4-aminophenoxy)phenyl]propane was thus confirmed to have been formed, as the appearance of these characteristic absorptions indicated that the reaction between bisphenol A and p-chloronitrobenzene had successfully produced BNPP and that BNPP had been completely reduced to the diamine monomer. In the ¹H-NMR spectrum, the aromatic proton signals of BNPP appeared in the region of 7.1–8.3 ppm, whereas 2,2-Bis[4-(4-aminophenoxy)phenyl]propane exhibited aromatic proton signals shifted to a lower field in the region of 6.5–7.2 ppm due to the inductive effect of the electron-withdrawing nitro groups, and the absorption corresponding to the amino group of the diamine monomer at 4.9 ppm further confirmed that the nitro groups had been fully reduced to amino groups. [1]
Synthesis of Polyimides
ODPA was added in several portions to a solution of 2,2-Bis[4-(4-aminophenoxy)phenyl]propane in DMAc, after which the mixture was stirred at 0 °C for 20 hours under an atmosphere of N₂ to facilitate the formation of the polyamic acid; 2,2-Bis[4-(4-aminophenoxy)phenyl]propane, serving as the diamine monomer in this polymerization, thus played a crucial role in the synthesis of the precursor polymer. The resulting polyamic acid solution was then cast onto a glass plate and subsequently subjected to a stepwise thermal imidization process, which involved heating at 80 °C for 2 hours, followed by 1 hour each at 120 °C, 180 °C, 250 °C, and finally 300 °C, during which DMAc was partially evaporated at 120 °C and completely removed at 180 °C owing to its boiling point of approximately 160 °C. 2,2-Bis[4-(4-aminophenoxy)phenyl]propane, as a key structural component of the resulting polyimide, contributed to the formation of a flexible polyimide film with an inherent viscosity of 1.3 dL/g. [1]
Reference
[1] Kong C, Zhang Q, Gu X, et al. Synthesis, structures and properties of polyimide based on 2, 2′‐bis (4‐aminophenoxy phenyl) propane[J]. Journal of Macromolecular Science, Part A: Pure and Applied Chemistry, 2006, 43(11): 1825-1833.
You may like
Related articles And Qustion
See also
Lastest Price from 2,2-Bis[4-(4-aminophenoxy)phenyl]propane manufacturers
![13080-86-9 2,2-Bis[4-(4-aminophenoxy)phenyl]propane](/ProductImageEN2/2026-03/Small/85f00902-264c-4ee4-93f5-4bd478023abf.png)
US $0.00-0.00/kg2026-03-25
- CAS:
- 13080-86-9
- Min. Order:
- 1kg
- Purity:
- 99.9%min
- Supply Ability:
- 20tons
![13080-86-9 2,2-Bis[4-(4-aminophenoxy)phenyl]propane](/ProductImageEN2/2025-12/Small/a0c4975e-d690-4c62-83e5-11e2ffff2a3a.jpg)
US $0.00-0.00/KG2025-12-23
- CAS:
- 13080-86-9
- Min. Order:
- 1KG
- Purity:
- 0.99
- Supply Ability:
- 10 tons
![13080-86-9 Preparation Method2,2-Bis[4-(4-aminophenoxy)phenyl]propane](https://www.chemicalbook.com/CAS/GIF/13080-86-9.gif)