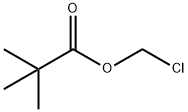
Preparation Methods and Application of Chloromethyl pivalate
Chloromethyl pivalate is a pivalate ester that exists as a clear, colorless to pale yellow liquid under ambient conditions. It is insoluble in water but miscible with most organic solvents. This compound can be synthesized via a condensation reaction between pivaloyl chloride and paraformaldehyde catalyzed by anhydrous zinc chloride. Chloromethyl pivalate serves as an intermediate in organic synthesis and pharmaceutical chemistry, and has been reported to be used in the preparation of drug molecules such as tebipenem pivoxil and adefovir pivoxil.
Preparation Methods
Method 1
Researchers have reported a simplified process for synthesizing chloromethyl pivalate. In this method, caprolactam is used as a catalyst to react pivalic acid with thionyl chloride at room temperature to generate pivaloyl chloride. Without isolation, an additional amount of thionyl chloride is added, and the mixture is then reacted directly with zinc chloride and paraformaldehyde under room‑temperature conditions to afford chloromethyl pivalate. The overall yield of chloromethyl pivalate reaches 94.2%, and the product structure is confirmed by IR and ¹H NMR. This approach is concluded to be a one‑step procedure with mild conditions, simple operation, and merits further development for industrial‑scale production.
Method 2
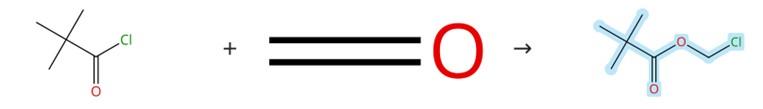
Figure1: Synthesis of Chloromethyl pivalate
Under a nitrogen atmosphere, a catalytic amount of ZnCl₂ was placed in a flask, followed by the addition of one equivalent of an acyl chloride (e.g., propionyl chloride). The mixture was cooled to between –5 °C and –10 °C, after which paraformaldehyde (1.38 g, 46 mmol) was introduced dropwise. The resulting reaction mixture was stirred at room temperature for one hour, then concentrated under reduced pressure. The crude product (e.g., chloromethyl propionate) was employed directly in the subsequent step without further purification. Similarly, chloromethyl pivalate was prepared from pivaloyl chloride and paraformaldehyde following the same general procedure, and the crude material was likewise used without additional purification. [2]
Application
Preparation of Sulbactam Pivoxil
Research has documented that chloromethyl pivalate serves as a key intermediate in the synthesis of the pharmaceutical compound sulbactam pivoxil. In the reported procedure, sulbactam sodium is reacted with chloromethyl pivalate in an organic medium—either N,N‑dimethylformamide or dimethyl sulfoxide—under phase‑transfer catalytic conditions maintained at 25 °C to 30 °C, affording sulbactam pivoxil as represented by formula (III). The phase‑transfer catalyst employed is a quaternary ammonium‑type cationic surfactant, while the molar ratio of sulbactam sodium to chloromethyl pivalate is controlled within the range of 1:1 to 1:1.5. This synthetic approach presents several distinct advantages: it establishes a novel pathway for the production of sulbactam pivoxil; the incorporation of a phase‑transfer catalyst facilitates high reaction yields under mild conditions; the operational steps are straightforward; the resulting product exhibits excellent quality; the overall process is cost‑effective; and the methodology is readily adaptable to large‑scale industrial manufacturing.
Etherification
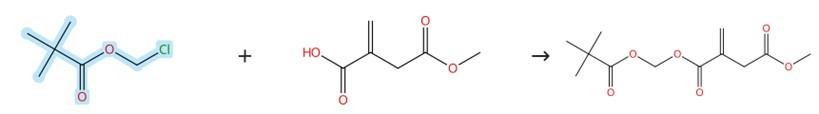
Figure2: Etherification of Chloromethyl pivalate
A mixture of 4‑methyl itaconate (200 mg), chloromethyl pivalate (272 mg, 260 μL), sodium iodide (41.6 mg, 0.278 mmol, 0.2 equiv) and potassium carbonate (288 mg, 2.08 mmol, 1.5 equiv) was dissolved in anhydrous acetonitrile (5 mL) and stirred at 40 °C for 16 hours. After completion, ethyl acetate (60 mL) was added, and the mixture was washed successively with brine (20 mL) and saturated sodium thiosulfate solution (10 mL). The organic phase was dried over anhydrous sodium sulfate, the volatiles were removed under reduced pressure, and the residue was purified by flash column chromatography on silica gel using cyclohexane/ethyl acetate (5:1) as eluent. [3]
Nucleophilic substitution reaction
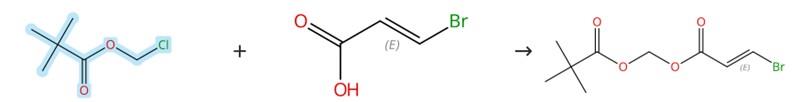
Figure3: Nucleophilic substitution reaction of Chloromethyl pivalate
To a solution of (2E)-3-bromoprop-2-enoic acid (1.51 g) in DMF (15 mL) were added triethylamine (2.63 mL, 18.9 mmol), chloromethyl pivalate (2.87 mL, 19.9 mmol), and sodium iodide (100 mg, 0.7 mmol). The resulting mixture was stirred at 70 °C for 15 min, then quenched with water and extracted with diethyl ether (3 × 20 mL). The combined organic layers were dried over sodium sulfate, filtered, and concentrated under reduced pressure. The residue was purified by normal-phase column chromatography (hexane/EtOAc = 30:1) to afford the desired product.[4]
References
[1] A New Synthetic Process for Chloromethyl Pivalate, Journal of Taishan Medical College, 2005, 26 (4): 2.
[2] J. Chen, Z.X. Liu, P. Wang et al., A Preparation Method for Sulbactam Pivoxil, Chinese Patent CN200910155773.6.
[3] C. B. Lee et al. Discovery of Orally Available Prodrugs of Itaconate and Derivatives, Journal of Medicinal Chemistry, 2025, 68, 3433?3444.
[4] Kotze, Timothy J.; et al, Metabolic Activation versus Masked Prodrugs: Bisubstrate Mimic Inhibitors of CoaBC's PPCS Activity in Mycobacterium tuberculosis and Staphylococcus aureus, ACS Infectious Diseases 2025, 11, 1508-1517.
You may like
Lastest Price from Chloromethyl pivalate manufacturers

US $10.00/KG2025-04-21
- CAS:
- 18997-19-8
- Min. Order:
- 1KG
- Purity:
- 99%
- Supply Ability:
- 10 mt

US $13.00/kg2025-04-18
- CAS:
- 18997-19-8
- Min. Order:
- 1kg
- Purity:
- 99.0%
- Supply Ability:
- 500MT