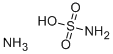Preparation Methods and Thermodynamic properties of Ammonium sulfamate
Ammonium sulfamate is an inorganic ammonium salt, which appears as a white, loose crystalline solid under standard temperature and pressure. It possesses a characteristic pungent odor and exhibits good chemical stability. This compound demonstrates certain solubility in water but is insoluble in petroleum ether. As a hazardous material, ammonium sulfamate can decompose explosively upon heating and may undergo spontaneous explosion in hot acidic solutions. When burned, it releases toxic gases such as nitrogen oxides and sulfur oxides. Due to its versatile properties, ammonium sulfamate is widely utilized across various industries including pesticides, dyeing, tobacco, construction materials, and textiles.

Figure1: Picture of Ammonium sulfamate
Physical characteristics
Regarding its physical characteristics, ammonium sulfamate typically presents as a white crystalline substance. It is soluble in water, yielding a weakly acidic solution—for instance, a 10% aqueous solution has a pH ranging from 4 to 6. However, it is insoluble in methanol and ethanol. Additionally, this compound is hygroscopic, meaning it can absorb moisture from the air. Chemically classified as a sulfur-containing ammonium salt, ammonium sulfamate serves as an effective flame retardant for polyamide 6 (PA6). It can be synthesized by neutralizing sulfamic acid with ammonia. In environmental contexts, ammonium sulfamate decomposes in soil to form ammonium sulfate and also exhibits notable herbicidal properties.
Thermodynamic properties
Molar heat capacity of Ammonium sulfamate (NH4SO3NH2) was measured in the temperature range from 8 K to 335 K by low-temperature vacuum adiabatic calorimetry. Obtained data were approximated by linear combination of Einstein functions. Heat content and entropy of Ammonium sulfamate were calculated from these data. Enthalpy of NH4SO3NH2 dissolution in water was determined at 298.15 K by means of solution calorimetry. On the basis of experimental data the standard entropy, enthalpy and Gibbs energy Ammonium sulfamate formation at 298.15 K were calculated. The phase transition of Ammonium sulfamate was observed by adiabatic calorimetry. [1]
Chemical Properties
Ammonium sulfamate is an inorganic compound that can be produced by the reaction of urea with sulfuric acid. At 25°C, its solubility in water is 216 g, and it is also soluble in formamide and glycerol. Ammonium sulfamate decomposes upon heating. The acute oral LD₅₀ of ammonium sulfamate in rats is 3900 mg/kg. Additionally, ammonium sulfamate functions as a non-selective herbicide and holds particular value in controlling most woody plants.
Preparation Methods
Under pressure, purified sulfamic acid is dissolved in liquid ammonia, followed by减压 (pressure reduction) to evaporate excess liquid ammonia, yielding the target product ammonium sulfamate. Literature also reports that ammonium sulfamate can be obtained by reacting sulfur trioxide with liquid ammonia under pressure. Moreover, ammonium sulfamate can be prepared through a metathesis reaction using ammonium bicarbonate and sulfamic acid as raw materials.
Protein Purification
Practical details of selective precipitation are presented in UNIT 4.5, and an example in the purification of interleukin 1β is given in UNIT 6.2. Salt precipitation has been widely used to fractionate membrane proteins (Schagger, 1994). Due to bound lipid and/or detergents, ammonium sulfate precipitates have lower density than protein-only precipitates. During centrifugation, these precipitates will often float to the top of tube rather than pelleting; the use of swing-out rotors is recommended. Crystallization is a traditional method of protein purification. Jakoby (1971) describes a general method that involves extracting Ammonium sulfamate-precipitated protein with successively dilute Ammonium sulfamate solutions at low temperature. [2]
Applications
Ammonium sulfamate serves as a non-selective herbicide in agricultural production. It is also widely used in industrial sectors such as pesticides, dyeing, tobacco, construction materials, and textiles. [3]
Nutrient source
In the agricultural sector, ammonium sulfamate is also an important nutritional additive. Rich in nitrogen, it serves as a key nutrient source for plants. Ammonium sulfamate promotes plant growth and development while improving crop yield and quality. Therefore, it is widely used in agricultural production, particularly in nitrogen-deficient soils, where it significantly enhances crop growth conditions.
Scientific research
Furthermore, ammonium sulfamate finds broad applications in chemical analysis and scientific research. It can be used as a standard substance and reagent in various analytical methods and experimental procedures. Its high purity and stability make it a preferred choice for researchers. In chemical studies, ammonium sulfamate is often employed in synthesizing new compounds and materials, with its reactivity and versatility making it central to numerous research projects.
Reference
[1] Kosova D A, Druzhinina A I, Tiflova L A, et al. Thermodynamic properties of ammonium sulfamate[J]. The Journal of Chemical Thermodynamics, 2019, 132: 432-438.
[2] Wingfield P. Protein precipitation using ammonium sulfate[J]. Current protocols in protein science, 1998, 13: 1.
[3] Yuan, S. Inorganic Herbicides [J]. Huaxue Shijie (Chemical World), 1994, (03): 162-163.
You may like
Lastest Price from Ammonium sulfamate manufacturers

US $10.00/KG2025-04-21
- CAS:
- 7773-06-0
- Min. Order:
- 1KG
- Purity:
- 99%
- Supply Ability:
- 10 mt

US $30.00-1.00/kg2025-03-07
- CAS:
- 7773-06-0
- Min. Order:
- 1kg
- Purity:
- 99
- Supply Ability:
- 20tons