Related Research on D-(+)-Cellobiose
Introduction
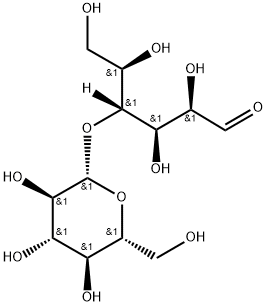
D-(+)-Cellobiose (Figure 1), a β-1,4-linked D-glucose disaccharide with a positive specific rotation, is a key intermediate in cellulose hydrolysis and a vital substrate for biomass biorefinery research. D-(+)-Cellobiose , which is the D-glucose dimer was chosen as a model compound for the initial phase of studies, as cellulose is insoluble in all common solvents and it is difficult to study the direct interactions of cellulose with acids in a heterogeneous phase.[1]

Interactions of D-(+)-Cellobiose with p-toluenesulfonic acid in aqueous solution
The effects of adding D2SO4, and p-toluenesulfonic acid-d to D-(+)-Cellobiose dissolved in D2O were investigated at 23°C by plotting (13)C NMR chemical shift changes (Δδ) against the acid to D-(+)-Cellobiose molar ratio. (13)C Chemical shifts of all 18 carbon signals from α and β anomers of D-(+)-Cellobiose showed gradual decreases due to increasing acidity in aqueous D2SO4 medium. The C-1 of the α anomer showed a slightly higher response to increasing D(+) concentration in the surrounding. In the aqueous p-toluenesulfonic acid-d medium, C-6' and C-4' carbons of both α, and β anomeric forms of D-(+)-Cellobiose are significantly affected by increasing the sulfonic acid concentrations, and this may be due to a 1:1 interaction of p-toluenesulfonic acid-d with the C-6', C-4' region of the cellobiose molecule.[1]
Interactions of D-(+)-Cellobiose with selected chloride salts
The interactions of cellulose model compound D-(+)-Cellobiose with chloride salts of Zn2+, Ca2+, Li+, Sn2+, La3+, Mg2+, K+ and NH4+ were evaluated by measuring the 13C NMR chemical shift changes (Δδ) of the disaccharide due to the addition of salts in D2O. The KCl and NH4Cl showed similar Δδ changes due to interactions only with the Cl- anion. Whereas other chloride salts showed interactions with both cation and anion. Among these salts the total interactions are in the order: Zn2+>Sn2+>Li+>Ca2+~La3+>Mg2+. The FT-IR spectra of D-(+)-Cellobiose-chloride salt 1:2 mixtures also indicate that KCl and NH4Cl interacts similarly with D-(+)-Cellobiose in the solid state.[2]
Effect of D-(+)-Cellobiose on oral bioavailability of gentiopicroside
In this study,the effect of D-(+)-Cellobiose on oral bioavailability of gentiopicroside ( GPS) was investigate. The influence of D-(+)-Cellobiose on GPS was achieved by calculating the residual GPS after being degraded with β-glucosidase or intestinal flora,and the data demonstrated D-(+)-Cellobiose could inhibit the degradation of GPS in intestines; in bioavailability experiment, D-(+)-Cellobiose could significantly improve the oral bioavailability ( P<0. 05) of GPS at the mass ratio of 1∶ 5,1∶ 10 ( GPS- D-(+)-Cellobiose) . D-(+)-Cellobiose applied in this study may improve the oral bioavailability of GPS through delaying the degradation in intestines.[3]
Recombinant Corynebacterium glutamicum under oxygen-deprived conditions
Corynebacterium glutamicum R was metabolically engineered to broaden its sugar utilization range to D-xylose and D-(+)-Cellobiose contained in lignocellulose hydrolysates. The resultant recombinants expressed Escherichia coli xylA and xylB genes, encoding D-xylose isomerase and xylulokinase, respectively, for D-xylose utilization and expressed C. glutamicum R bglF317A and bglA genes, encoding phosphoenolpyruvate:carbohydrate phosphotransferase system (PTS) beta-glucoside-specific enzyme IIBCA component and phospho-beta-glucosidase, respectively, for D-(+)-Cellobiose utilization. The genes were fused to the non-essential genomic regions distributed around the C. glutamicum R chromosome and were under the control of their respective constitutive promoter trc and tac that permitted their expression even in the presence of D-glucose. The enzyme activities of resulting recombinants increased with the increase in the number of respective integrated genes. Maximal sugar utilization was realized with strain X5C1 harboring five xylA-xylB clusters and one bglF317A-bglA cluster. In both D-(+)-Cellobiose and D-xylose utilization, the sugar consumption rates by genomic DNA-integrated strain were faster than those by plasmid-bearing strain, respectively. In mineral medium containing 40 g l-1 D-glucose, 20 g l-1 D-xylose, and 10 g l-1 D-(+)-Cellobiose, strain X5C1 simultaneously and completely consumed these sugars within 12 h and produced predominantly lactic and succinic acids under growth-arrested conditions.[4]
Hydrolysis and interactions of D-(+)-Cellobiose with polycarboxylic acids
The hydrolysis of cellulose model compound D-(+)-Cellobiose was studied with a series of eight common polycarboxylic acids and two monocarboxylic acids in aqueous medium using 0.500 mmol -COOH/L at 170 °C. The maleic acid showed the highest catalytic activity with turnover frequency (TOF) of 29.5 h -1. The interaction of carboxylic acids with D-(+)-Cellobiose in DMSO-d 6 was studied by determination of the pseudo first-order rate constant k H of anomeric -OH exchange rate in cellobiose using 1H NMR spectroscopy. The maleic, oxalic and citric acids showed infinitely large k H values indicating very strong interactions with D-(+)-Cellobiose. The next highest interactions were found with phthalic acid (kH = 248.8 Hz). The FT-IR studies showed significant carboxylic acid C=O stretching frequency shifts (ΔνC=O) of 12, 13 and 10 cm-1 for maleic, oxalic and acetic acids respectively in mixtures with D-(+)-Cellobiose.[5]
References
[1] Amarasekara AS, Owereh OS, Ezeh B. Interactions of D-cellobiose with p-toluenesulfonic acid in aqueous solution: a (13)C NMR study. Carbohydr Res. 2011;346(17):2820-2822. doi:10.1016/j.carres.2011.09.032
[2] Amarasekara AS, Wiredu B. Interactions of D-cellobiose with selected chloride salts: A ¹³C NMR and FT-IR study. Spectrochim Acta A Mol Biomol Spectrosc. 2016;159:113-116. doi:10.1016/j.saa.2016.01.048
[3] Dai YH,et al.Effect of D-cellobiose on oral bioavailability of gentiopicroside[J].China Journal of Chinese Materia Medica,2016,41(10):1855-1859.
[4] Sasaki M, Jojima T, Inui M, Yukawa H. Simultaneous utilization of D-cellobiose, D-glucose, and D-xylose by recombinant Corynebacterium glutamicum under oxygen-deprived conditions. Appl Microbiol Biotechnol. 2008;81(4):691-699. doi:10.1007/s00253-008-1703-z
[5] Amarasekara AS, Wiredu B, Lawrence YM. Hydrolysis and interactions of d-cellobiose with polycarboxylic acids. Carbohydr Res. 2019;475:34-38. doi:10.1016/j.carres.2019.02.002
You may like
See also
Lastest Price from D-(+)-Cellobiose manufacturers

US $0.00-0.00/kg2025-12-05
- CAS:
- 528-50-7
- Min. Order:
- 1kg
- Purity:
- 98%
- Supply Ability:
- 1000kg

US $0.00/KG2025-04-21
- CAS:
- 528-50-7
- Min. Order:
- 1KG
- Purity:
- 98%min
- Supply Ability:
- 30tons/month