Related Research on Pentaerythritol Triacrylate: Applications and Toxicity
Introduction
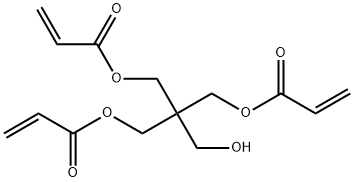
Pentaerythritol triacrylate (Figure 1) is used in the production of ultraviolet-curable inks and coatings, electron beam irradiation-curable coatings, and radiation-cured and photocurable coatings of urethanes and epoxy resins; as a component of photopolymer and flexographic printing inks and plates and photoresists; as an ingredient of acrylic glues, adhesives, and anaerobic sealants; and as a modifier for polyester and fiberglass. It is also used in colloidal dispersions for industrial baked coatings,waterborne and solvent-based alkyds, vinyl/acrylic nonwoven binders, paper and wood impregnates, wire and cable extrusion, polymer-impregnated concrete, and polymer concrete structural composites. Pentaerythritol triacrylate was nominated by the National Cancer Institute for testing based on its high production volume and use, its potential for human exposure, and a lack of adequate testing of the chemical. [1]

Genetic toxicology
Pentaerythritol triacrylate was not mutagenic in several strains of S. typhimurium, with or without hamster or rat liver S9 activation enzymes. No increase in the frequency of micronucleated erythrocytes was observed in peripheral blood samples from B6C3F1 mice treated with pentaerythritol triacrylate by skin painting for3 months. In contrast, similar treatment of female Tg.AC hemizygous mice for 6 months induced a significant increase in micronucleated erythrocytes; the increase in micronuclei seen in male Tg.AC hemizygous mice was judged to be equivocal.
Contact hypersensitivity studies
Studies were conducted with female BALB/c mice to evaluate the potential for pentaerythritol triacrylate to induce contact hypersensitization. In an irritancy study in which formulations of pentaerythritol triacrylate(approximately 10% or 45% pure) in acetone were applied to the ear, the maximal nonirritating concentration was 0.1% and the minimal irritating concentration was 0.25% for both mixtures. A mouse ear swelling test yielded negative results for pentaerythritol triacrylate as a potential contact sensitizer using the 10% mixture and positive results with the 45% mixture. Positive responses were seen in local lymph node assays at concentrations of 0.05%, 0.1%, and 0.25% pentaerythritol triacrylate when the approximately 10% pentaerythritol triacrylate mixture was used and at a concentration of0.25% pentaerythritol triacrylate when the approximately 45% pentaerythritol triacrylate mixture was used.
In summary, male and female Tg.AC hemizygous mice dosed with pentaerythritol triacrylate for 6 months had significantly increased incidences of squamous cell papillomas of the skin at the site of dermal application. Treatment-related squamous cell carcinomas occurred at the site of application in male mice. Nonneoplastic lesions noted at the site of application included hyperkeratosis, chronic active inflammation, and epidermal hyperplasia. A hematopoietic disorder (myelodysplasia) occurred in dosed male mice.[1]
Highly cross-linked networks for dental applications
Highly cross-linked networks have been obtained by photoinitiated camphorquinone-amine polymerization of tris[2-(acryloyloxy)ethyl]isocyanurate alone and/or with 2-ethyl-2-(hydroxymethyl)-1,3-propanediol triacrylate or pentaerythritol triacrylate, which can be considered a new class of dental restorative resins. The most effective coinitiators in this system are amines such as ethyl-4-dimethylaminobenzoate, N,N-dimethyl-benzylamine, and 2,4,6-tris(dimethylaminomethyl)phenol. The volume shrinkages of polymerized samples were 8%-13%. The hardness of photocured resins in the presence of an inorganic filler (aluminum/fluoro/silicate glass, Ketac-Fil was slightly less than that of a restorative composite material (Z100 MP).[2]
Characterization of pentaerythritol triacrylate-co-trimethylolpropane nanocomposite scaffolds
A thiol-acrylate-based copolymer synthesized via an amine-catalyzed Michael addition was studied in vitro and in vivo to assess its potential as an in situ polymerizing graft or augment in bone defect repair. The blends of hydroxyapatite (HA) with pentaerythritol triacrylate-co-trimethylolpropane (PETA), cast as solids or gas foamed as porous scaffolds, were evaluated in an effort to create a biodegradable osteogenic material for use as a bone-void-filling augment. Osteogenesis experiments were conducted with human adipose-derived mesenchymal stromal cells (hASCs) to determine the ability of the material to serve as an osteoinductive substrate. Poly(ɛ-caprolactone) (PCL) composites PCL:HA (80:20) (wt/wt%) served as the control scaffold, while the experimental scaffolds included PETA:HA (100:0), (85:15), (80:20), and (75:25) composites (wt/wt%). The results indicate that PETA:HA (80:20) foam composites had higher mechanical strength than the corresponding porous PCL:HA (80:20) scaffolds made by thermo-precipitation method, and in the case of foamed composites, increasing HA content directly correlated with increased yield strength. For cytotoxicity and osteogenesis experiments, hASCs cultured for 21 days on PETA:HA scaffolds in stromal medium displayed the greatest number of live cells compared with PCL:HA composites. Moreover, hASCs cultured on foamed PETA:HA (80:20) scaffolds resulted in the greatest mineralization, increased alkaline phosphatase (ALP) expression, and the highest osteocalcin (OCN) expression after 21 days. Overall, the PETA:HA (80:20) and PETA:HA (85:15) scaffolds, with 66.38% and 72.02% porosity, respectively, had higher mechanical strength and cytocompatibility compared with the PCL:HA control. The results of the 6-week in vivo biocompatibility study using a posterior lumbar spinal fusion model demonstrate that PETA:HA can be foamed in vivo without serious adverse effects at the surgical site. Additionally, it was demonstrated that cells migrate into the interconnected pore volume and are found within centers of ossification.[3]
Catalytically Active Light Printed Microstructures
Light-induced additive manufacturing (3D printing) has revolutionized manufacturing and its integration into the fabrication of catalysts holds key potential to enable facile access to optimized catalyst geometries and designs. Herein - for the first time - micro- and macro-sized photocatalytically active 3D printed objects are introduced via a dual-function photoresin using a ruthenium(II) complex containing monomer as both a photoinitiator for the 3D printing process and as the active photocatalyst within the printed structure. The approach leverages the spatial and temporal control afforded by light-induced 3D printing techniques during both one- and two-photon printing to precisely position the photocatalyst within intricate geometries using a pentaerythritol triacrylate (PETA) based resin. The successful incorporation of ruthenium(II) complexes is demonstrated via time-of-flight secondary-ion mass spectrometry (ToF-SIMS) into desired sections of 3D-printed objects. The one- and two-photon fabricated architectures show photocatalytic activity in the C─H arylation of activated aryl bromides. The potential of tailored catalytically active 3D objects is exemplified by one of the microscale designs. This design, utilizing only 1% of the volume of a macroscale structure fabricated from the same resin, achieved 75% of the photocatalytic performance.[4]
References
[1] National Toxicology Program. Toxicology studies of pentaerythritol triacrylate (technical grade) (CAS No. 3524-68-3) in F344/N rats, B6C3F1 mice, and genetically modified (FVB Tg.AC hemizygous) mice (dermal studies). Natl Toxicol Program Genet Modif Model Rep. 2005;(4):1-190.
[2] Nie J, Lindén LA, Rabek JF, Ekstrand J. Highly cross-linked networks for dental applications obtained by photocuring of tris[2-(acryloyloxy)ethyl]isocyanurate, 2-ethyl-2-(hydroxymethyl)-1,3-propanediol triacrylate, and pentaerythritol triacrylate. Acta Odontol Scand. 1999;57(1):1-8. doi:10.1080/000163599429039
[3] Chen C, Garber L, Smoak M, et al. In vitro and in vivo characterization of pentaerythritol triacrylate-co-trimethylolpropane nanocomposite scaffolds as potential bone augments and grafts. Tissue Eng Part A. 2015;21(1-2):320-331. doi:10.1089/ten.TEA.2014.0018
[4] Finch AK, Gillhuber S, Frisch H, Roesky PW, Barner-Kowollik C. Catalytically Active Light Printed Microstructures. Adv Mater. 2025;37(34):e2506663. doi:10.1002/adma.202506663
You may like
Lastest Price from Pentaerythritol triacrylate manufacturers

US $0.00-0.00/kg2025-06-13
- CAS:
- 3524-68-3
- Min. Order:
- 1kg
- Purity:
- 99.0% min
- Supply Ability:
- 10tons

US $1.00/KG2025-04-21
- CAS:
- 3524-68-3
- Min. Order:
- 1KG
- Purity:
- 99%
- Supply Ability:
- 10mt