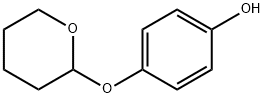
Research on the Effects of Deoxyarbutin
Introduction
Deoxyarbutin (4-[(tetrahydro-2H-pyran-2-yl) oxy] phenol, dA) (Fig. 1), a commercial product in skin lightening, appears to have similar activities as hydroquinone (1, 4-benzenediol, HQ). Previous studies have demonstrated HQ could inhibit tyrosine activity as well as induce DNA damage via generation of reactive oxygen species (ROS). Wang et al. showed HQ could induce apoptosis and result in cytotoxicity in mouse primary hepatocytes, which could be reserved by resveratrol. Hydroxyl hydroquinone (HHQ), also obtained from HQ, was observed to decrease cell viability and colony formation by induction of oxidative stress that leads to apoptosis. Deoxyarbutin was safer and less cytotoxic compared with HQ and HHQ. Miao et al. revealed that deoxyarbutin possessed a potent skin lightening ability by regulating ROS generation with less melanosome cytotoxicity in vitro and in vivo models.[1]

Deoxyarbutin displays antitumour activity against melanoma in vitro and in vivo
Apoptosis is a major route to deracinate cancer cells and involves cell cycle arrest. Melanoma is a disease characterized by high lung metastases. In this study, Ma etal. firstly provided that deoxyarbutin could significantly inhibit the growth of melanoma cells in vitro and B16F10 xenografts as well as pulmonary metastasis in vivo. These results also elaborated detailed mechanistic of deoxyarbutin’s antitumour action on melanoma cancer (mitochondrial dysfunction,caspases-family activation).Furthermore, deoxyarbutin triggered its pro-apoptosis through damaging the mitochondrial function (membrane potential loss, ATP depletion and ROS overload generation etc.) and activating caspase-9, PARP, caspase-3 and the phosphorylation of p38. Treatment with p38 agonist confirmed the involvement of p38 pathway triggered by deoxyarbutin in B16F10 cells. The in vivo finding also revealed that administration of deoxyarbutin significantly decreased the tumour volume and tumour metastasis in B16F10 xenograft model by inhibiting tumour proliferation and inducing tumour apoptosis. Importantly, the results indicated that deoxyarbutin was specific against tumour cell lines and had no observed systemic toxicity in vivo. Taken together, these study demonstrated that deoxyarbutin could combate tumour in vitro and in vivo by inhibiting the proliferation and metastasis of tumour via a p38-mediated mitochondria associated apoptotic pathway.[1]
Deoxyarbutin targets mitochondria to trigger p53-dependent senescence of glioblastoma cells
Cellular senescence is a natural barrier of the transition from premalignant cells to invasive cancer. Pharmacological induction of senescence has been proposed as a possible anticancer strategy. In this study, Cai etal. found that deoxyarbutin inhibited the growth of glioblastoma (GBM) cells by inducing cellular senescence, independent of tyrosinase expression. Instead, deoxyarbutin induced mitochondrial oxidative stress and damage. These aberrant mitochondria were key to the p53-dependent senescence of GBM cells. Facilitating autophagy or mitigating mitochondrial oxidative stress both suppressed p53 expression and alleviated cellular senescence induced by deoxyarbutin. Thus, our study reveals that deoxyarbutin induces mitochondrial oxidative stress to trigger the p53-dependent senescence of GBM cells. Importantly, deoxyarbutin treatment resulted in accumulation of p53, induction of cellular senescence, and inhibition of tumor growth in a subcutaneous tumor model of mouse. In conclusion, our study reveals that deoxyarbutin has therapeutic potential for GBM by inducing mitochondrial oxidative stress for p53-dependent senescence of GBM cells.[2]
Deoxyarbutin attenuates severe acute pancreatitis via the HtrA2/PGC-1alpha pathway
Severe acute pancreatitis (SAP) is an inflammatory disorder of the exocrine pancreas associated with high morbidity and mortality. SAP has been proven to trigger mitochondria dysfunction in the pancreas. Li etal. found that Deoxyarbutin (dA) recovered impaired mitochondrial function. High-temperature requirement protein A2 (HtrA2), a mitochondrial serine protease upstream of PGC-1α, is charge of quality control in mitochondrial homeostasis. The molecular docking study indicated that there was a potential interaction between deoxyarbutin and HtrA2. However, whether the protective effect of deoxyarbutin against SAP is regulated by HtrA2/PGC-1α remains unknown. These study in vitro showed that deoxyarbutin significantly reduced the necrosis of primary acinar cells and reactive oxygen species (ROS) accumulation, recovered mitochondrial membrane potential (ΔΨm) and ATP exhaustion, while UCF-101 (HtrA2 inhibitor), and SR-18292 (PGC-1α inhibitor) eliminated the protective effect of deoxyarbutin. Moreover, HtrA2 siRNA transfection efficiently blocked the protective of deoxyarbutin on HtrA2/PGC-1α pathway in 266-6 acinar cells. Meanwhile, deoxyarbutin also decreased LC3II/I ration, as well as p62, and increased Parkin expression, while UCF-101 and Bafilomycin A1 (autophagy inhibitor) reversed the protective effect of deoxyarbutin. Our study in vivo confirmed that dA effectively alleviated severity of SAP by reducing pancreatic edema, plasma amylase, and lipase levels and improved the HtrA2/PGC-1α pathway. Therefore, this is the first study to identify that deoxyarbutin inhibits pancreatic injury caused by oxidative stress, mitochondrial dysfunction, and impaired autophagy in a HtrA2/PGC-1α dependent manner.[3]
Structural and kinetic considerations on the catalysis of deoxyarbutin by tyrosinase
Deoxyarbutin, a potent inhibitor of tyrosinase, could act as substrate of the enzyme. Oxytyrosinase is able to hydroxylate deoxyarbutin and finishes the catalytic cycle by oxidizing the formed o-diphenol to quinone, while the enzyme becomes deoxytyrosinase, which evolves to oxytyrosinase in the presence of oxygen. This compound is the only one described that does not release o-diphenol after the hydroxylation step. Oxytyrosinase hydroxylates the deoxyarbutin in ortho position of the phenolic hydroxyl group by means of an aromatic electrophilic substitution. As the oxygen orbitals and the copper atoms are not coplanar, but in axial/equatorial position, the concerted oxidation/reduction cannot occur and the release of a copper atom to bind again in coplanar position, enabling the oxidation/reduction or release of the o-diphenol from the active site to the medium. In the case of deoxyarbutin, the o-diphenol formed is repulsed by the water due to its hydrophobicity, and so can bind correctly and be oxidized to a quinone before being released. Deoxyarbutin has been characterized with: Kcat=1.95±0.06 s-1 and KM=33±4μM. Computational simulations of the interaction of β-arbutin, deoxyarbutin and their o-diphenol products with tyrosinase show how these ligands bind at the copper centre of tyrosinase. The presence of an energy barrier in the release of the o-diphenol product of deoxyarbutin, which is not present in the case of β-arbutin, together with the differences in polarity and, consequently differences in their interaction with water help understand the differences in the kinetic behaviour of both compounds. Therefore, it is proposed that the release of the o-diphenol product of deoxyarbutin from the active site might be slower than in the case of β-arbutin, contributing to its oxidation to a quinone before being released from the protein into the water phase.[4]
References
[1] Ma L, Xu Y, Wei Z, et al. Deoxyarbutin displays antitumour activity against melanoma in vitro and in vivo through a p38-mediated mitochondria associated apoptotic pathway. Sci Rep. 2017;7(1):7197. Published 2017 Aug 3. doi:10.1038/s41598-017-05416-8
[2] Cai D, Xu X, Zeng W, et al. Deoxyarbutin targets mitochondria to trigger p53-dependent senescence of glioblastoma cells. Free Radic Biol Med. 2024;224:382-392. doi:10.1016/j.freeradbiomed.2024.08.027
[3] Li Y, Zhu Y, Li S, et al. Deoxyarbutin attenuates severe acute pancreatitis via the HtrA2/PGC-1α pathway. Free Radic Res. 2022;56(9-10):651-665. doi:10.1080/10715762.2022.2163244
[4] Garcia-Jimenez A, Teruel-Puche JA, Garcia-Ruiz PA, et al. Structural and kinetic considerations on the catalysis of deoxyarbutin by tyrosinase. PLoS One. 2017;12(11):e0187845. Published 2017 Nov 14. doi:10.1371/journal.pone.0187845
You may like
Lastest Price from Deoxyarbutin manufacturers
US $0.00-0.00/kg2025-09-08
- CAS:
- 53936-56-4
- Min. Order:
- 1kg
- Purity:
- 98%
- Supply Ability:
- 1

US $0.00/KG2025-08-30
- CAS:
- 53936-56-4
- Min. Order:
- 1KG
- Purity:
- 99%
- Supply Ability:
- 2000KG