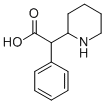
Ritalinic Acid: a Methylphenidate Metabolite
Ritalinic acid is defined as a metabolite of methylphenidate (MPH), which is rapidly produced during its metabolism in the body. It is formed through hydrolysis or deesterification of methylphenidate in the body and is pharmacologically inactive. Ritalinic acid can be identified and quantified in serum as a way to monitor the use of methylphenidate and related drugs.

Ritalinic Acid Stimulates Human Sperm Motility and Maintains Vitality In Vitro
Attention-deficit hyperactivity disorder (ADHD) is a frequent neurodevelopmental disorder with an increasing prevalence over the last 2 decades. A recent cohort study of almost 7.5 million people with roughly 700,000 prescriptions, reported an increase in prescription rate from 42.7 prescriptions per 10,000 persons before the year 2000 to 394.4 prescriptions in 2015 an increase of about 800% in prescription rate. The upsurge was observed in both sexes and in all age groups. Ritalinic acid (RA), the major inactive metabolite of MPH, is one of the isomers of amphetamine. The circulating concentrations of RA greatly exceed that of the parent drug. If MPH is given in the normal dosage for clinical use, the plasma concentration of RA reaches a maximal concentration of 10 ng/mL with a half-life of 2.5 hours. Ritalinic acid is soluble in water and due to its small size, it readily passes the blood brain and testicular barriers. However, no study assessed the effect of MPH or its metabolites on human sperm. Nonetheless, the growing use of MPH in adolescents and adults who are in their reproductive age, and the growing rate of MPH abuse, requires an evaluation of the impact of MPH on male reproduction. The aim of the present study is to investigate the direct effect of Ritalinic acid on human semen parameters in vitro.[1]
Results of the current study clearly show that MPH main metabolite, Ritalinic acid, was able to increase human sperm motility, while sperm kinematic parameters, such as LIN, ALH, and VCL were not affected. In addition, vitality was maintained over time at significantly higher levels as compared to the control. It is also important to note that despite increased oxidative stress (OS) levels as measured as ORP after 240 and 300 minutes, respectively, these increases were not significant. Moreover, the impact of RA on sperm functions was comparable in all studied RA concentrations including the subclinical dose of 1 ng/mL, and the abuse equivalent doses of 100 and 1,000 ng/mL. However, due to the fact that the actual Ritalinic acid concentrations in semen have never been reported before, concentrations used in this study were only based on plasma levels and has to be considered as a limitation of our study. In conclusion, RA significantly increased sperm motility and maintained vitality over a long incubation period. This may allow its in vitro use to improve sperm motility in cases of patients with asthenozoospermia. However, it is unknown what effects Ritalinic acid has on other sperm functional characteristics such as: capacitation, acrosome reaction, intrinsic ROS production and sperm nuclear DNA fragmentation. Further investigation is required prior to considering its clinical use.
Stability and uptake of methylphenidate and ritalinic acid in nine-spine stickleback
The presence of pharmaceuticals in the environment has drawn widespread concern from researchers and environmental managers over the impacts that exposure to pharmaceuticals might have on, for example, antibiotic resistance and aquatic wildlife. Many pharmaceutical compounds are present in the environment, but attention on compounds that modulate human behaviour—such as antidepressants, anxiolytics, or stimulants—has recently grown. Methylphenidate increases dopaminergic and norepinephric transmission via inhibiting the reuptake of these catecholamines from the synaptic cleft. Following a standard dose (18 mg), up to 80% is excreted as ritalinic acid (α-phenyl-2-piperidine acetic acid) that has little pharmacological activity. Given recent and growing research attention on methylphenidate and ritalinic acid as environmental pollutants, more detailed investigations of their stability in the water column and concentrations in the tissues of aquatic organisms are needed. Basic data on the stability of compounds in water and uptake in tissue is needed to effectively design laboratory tests. To support future studies on effects on aquatic organisms, we measured concentrations of methylphenidate and ritalinic acid in the water column under static laboratory conditions, with and without the presence of biota over 2 weeks.[2]
To support future ecotoxicology studies on these compounds, we assessed the degradation of methylphenidate and formation of ritalinic acid in the water column and tissue uptake in two aquatic organisms at two temperatures. The studies were performed well below the pKa of methylphenidate to avoid issues on ionization that has an impact on tissue uptake (Armitage et al. 2013) but also on adsorption processes (Rybacka and Andersson 2016). Methylphenidate hydrolyzed to ritalinic acid in the water column faster at 20 °C than 10 °C because higher temperatures increase the rate of the physicochemical processes leading to the transformation. In contrast, water louse did not reach peak methylphenidate concentrations until 5–7 days following the initial exposure, depending on temperature. This indicates that water louse may have different rates or mechanisms of uptake and/or different means of metabolizing or eliminating methylphenidate. In humans, methylphenidate is metabolized to this acid primarily by carboxylesterase CES1A1. In conclusion, we have provided the first analysis of the degradation and uptake of methylphenidate and ritalinic acid under controlled conditions in a geographically widespread aquatic vertebrate and invertebrate. Generally, methylphenidate appears to be of greater concern for aquatic invertebrates based on tissue uptake patterns.
Ritalinic acid in urine: Impact of age and dose
Urine drug testing (UDT) is often employed to help assess patient adherence to chronic drug prescriptions. Since 80% of the oral dose of MP is excreted in urine as ritalinic acid (RA), the primary metabolite of MP, the resulting concentrations of RA can be relatively high making identification of diversion and other abuse pathways difficult in the absence of historical metabolite levels from a normal patient population. Box and whiskers plots of the Ritalinic acid data were curated as discussed in the methods section in an attempt to define “normal” ranges of RA from MP patients. While the overall range (all data) is interesting, the box and whiskers plots representing school age patients and adult patients demonstrate the difference between these unique populations. It is clear that these two populations have statistically significant different median values with the school age patients exhibiting the highest concentrations. A close look at does not clearly indicate any correlation between these average/median values and body weight, creatinine, or daily dose. School age patients exhibit a median dose of 27 mg/day while adult patients have a median dose of 20 mg/day, a ratio of 1.35.[3]
While an increasing number of children and young adults continue to be dosed with MP as well as other stimulants for the treatment of ADHD, little has been written about testing concentrations of Ritalinic acid in urine and what is “normal” vs. what is diversion/abuse. These higher dose levels coupled with lower body weight in this age group might account for a portion of the observed differences. A more interesting question concerns the elevated dose levels for children vs adults. The observed differences between these groups cannot easily be attributed to a single factor or even a small collection of factors. Finally, if a UDT concentration is outside "normal" ranges, other clinical information/observations should guide decision making.
References
[1]Harlev A, Henkel R, Samanta L, Agarwal A. Ritalinic Acid Stimulates Human Sperm Motility and Maintains Vitality In Vitro. World J Mens Health. 2020 Jan;38(1):61-67. doi: 10.5534/wjmh.180127. Epub 2019 May 8. PMID: 31081298; PMCID: PMC6920070.
[2]McCallum ES, Lindberg RH, Andersson PL, Brodin T. Stability and uptake of methylphenidate and ritalinic acid in nine-spine stickleback (Pungitius pungitius) and water louse (Asellus aquaticus). Environ Sci Pollut Res Int. 2019 Mar;26(9):9371-9378. doi: 10.1007/s11356-019-04557-9. Epub 2019 Feb 25. PMID: 30805842; PMCID: PMC6469618.
[3]Feng S, Strickland E, Enders J, Roslawski M, McIntire T, McIntire G. Ritalinic acid in urine: Impact of age and dose. Pract Lab Med. 2021 Oct 23;27:e00258. doi: 10.1016/j.plabm.2021.e00258. PMID: 34754895; PMCID: PMC8561308.
You may like
See also
Lastest Price from Ritalinic acid manufacturers

US $0.00-0.00/kg2026-01-19
- CAS:
- 19395-41-6
- Min. Order:
- 1kg
- Purity:
- 99%
- Supply Ability:
- 20MT

US $0.00-0.00/kg2026-01-09
- CAS:
- 19395-41-6
- Min. Order:
- 1kg
- Purity:
- 99%min
- Supply Ability:
- 100 KG