Scalable Synthesis and Condensation reaction of 3,5-Dichlorobenzoic acid
3,5-Dichlorobenzoic acid is an organochlorine compound and one of the six isomers of dichlorobenzoic acid. It can be reacted with oxalyl chloride in dichloromethane using DMF as a catalyst to produce 3,5-dichlorobenzoyl chloride. Due to the electron-withdrawing effects of its two chlorine atoms, 3,5-dichlorobenzoic acid exhibits stronger acidity than benzoic acid itself. In the field of organic synthesis, 3,5-dichlorobenzoic acid is often used in the preparation of transition metal ion complexes and has promising applications in the synthesis of such coordination compounds.
Scalable Synthesis
As a crucial intermediate that finds extensive applications across the pharmaceutical, agrochemical, and broader chemical industries, 3,5-Dichlorobenzoic acid is conventionally synthesized using established methods; however, these traditional approaches typically necessitate harsh reaction conditions that not only exhibit poor regioselectivity but also raise significant environmental concerns, prompting the development of a novel three-step synthetic approach in this study that starts from commercially available and inexpensive p-chlorobenzoic acid and has been specifically designed for the preparation of 3,5-Dichlorobenzoic acid without the need for any chromatographic purification steps. This optimized synthetic route begins with an efficient amination reaction to form p-aminobenzoic acid, which is then subjected to a selective dichlorination process that is precisely guided by the directing effects of the amino group, and the sequence ultimately concludes with a mild diazo deamination reaction to yield the final product. Throughout this entire process, 3,5-Dichlorobenzoic acid is obtained with notably high purity and a total yield exceeding 60%, demonstrating significant potential for industrial production and offering an optimal green synthetic route for this key intermediate. [1]
Synthesis of lanthanide complexes
3,5-Dichlorobenzoic acid was employed as the primary ligand in the solvothermal synthesis of two novel lanthanide complexes, [La₃(L)₈(phen)(OH)(H₂O)]ₙ (1) and [Dy(L)₃(EtOH)]ₙ (2), with phenanthroline used as an auxiliary ligand where applicable. The synthesized complexes were characterized using single-crystal X-ray diffraction, infrared spectroscopy, elemental analysis, powder X-ray diffraction, and thermogravimetric analysis. In complex 1, 3,5-dichlorobenzoic acid contributes to the formation of one-dimensional chains featuring unique hexanuclear La₆ units, which are further extended into a three-dimensional supramolecular network through π–π stacking and hydrogen-bonding interactions. In complex 2, 3,5-dichlorobenzoic acid facilitates the construction of one-dimensional dysprosium chains, which are bridged by double and quadruple carboxylate groups from the ligand and assembled into a two-dimensional layer via weak hydrogen bonds. Magnetic studies revealed that complex 2 exhibits antiferromagnetic behavior. In the formation of complex 1, 3,5-Dichlorobenzoic acid links La₄ tetramers through carboxyl oxygen atoms to generate distinctive hexanuclear La₆ units. These units are further arranged by 3,5-Dichlorobenzoic acid into an extended one-dimensional lanthanum chain. For complex 2, 3,5-Dichlorobenzoic acid acts as a bridge to construct one-dimensional dysprosium chains. [2]
Condensation reaction
The carboxylic acid unit within the structure of 3,5-dichlorobenzoic acid exhibits the general physicochemical properties typical of alkyl carboxylic acids, enabling it to undergo condensation reactions with aniline derivatives in the presence of condensing agents to yield the corresponding amide derivatives.
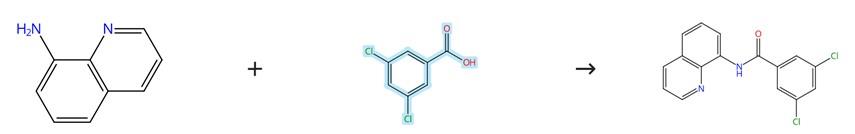
Figure1: Condensation reaction of 3,5-Dichlorobenzoic acid
To initiate the synthesis, a round-bottom flask equipped with a magnetic stir bar is sequentially charged with 3,5-Dichlorobenzoic acid (1.0 equivalent), 8-aminoquinoline (1 mmol, 1.0 equivalent), DMAP (1 mmol, 1.0 equivalent), and DCC (1.5 mmol, 1.5 equivalent), after which dichloromethane is added to achieve a 0.3 M concentration, and the resulting reaction mixture is allowed to stir continuously at room temperature for a period of 24 hours while the progress of the reaction is meticulously monitored by thin-layer chromatography until completion. Upon full conversion of the starting materials, the mixture is filtered through filter paper to remove insoluble by-products, and the filtrate is subsequently concentrated by removing the solvent under reduced pressure. The resulting crude product is then purified by flash column chromatography, utilizing a suitable mixture of ethyl acetate and n-hexane as the eluent, to afford the pure target compound, 3,5-dichloro-N-(quinolin-8-yl)benzamide. [3]
Reference
[1] Li S, Xue W, Chen Y, et al. An Efficient and Scalable Approach to the Synthesis of 3, 5-Dichlorobenzoic Acid[J]. Reaction Chemistry & Engineering, 2026.
[2] Tan X, Che Y X, Zheng J M. Syntheses, structures and characterizations of two Ln (III)-3, 5-dichlorobenzoate coordination polymers[J]. Inorganic Chemistry Communications, 2013, 35: 231-234.
[3] Mishra, Priyanka; et al, Nickel-Catalyzed Remote C(sp2)-H Nitration of 8-Aminoquinolines Using AgNO3 in Acetone, ChemistrySelect 2025, 10, e02763.
You may like
Related articles And Qustion
See also
Lastest Price from 3,5-Dichlorobenzoic acid manufacturers

US $0.00-0.00/kg2025-11-19
- CAS:
- 51-36-5
- Min. Order:
- 1kg
- Purity:
- 98%
- Supply Ability:
- 100tons

US $10.00/ASSAYS2025-08-21
- CAS:
- 51-36-5
- Min. Order:
- 1ASSAYS
- Purity:
- 99%
- Supply Ability:
- 1 ton