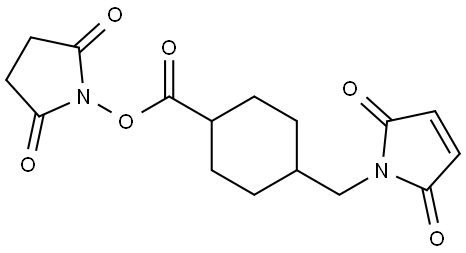
SMCC Crosslinker: Chemistry and Conjugation Applications
SMCC crosslinker is a heterobifunctional crosslinker, possessing two distinct reactive groups: an N-hydroxysuccinimide (NHS) ester and a maleimide. This dual reactivity enables a controlled, two-step conjugation process. First, the NHS ester reacts with primary amines, such as the lysine residues on the surface of the antibody, to form a stable amide bond. This reaction is typically carried out in a slightly alkaline pH range (7-9). Following this initial activation of the antibody, the maleimide group of the linker is then available to react specifically with a thiol (sulfhydryl) group present on the cytotoxic payload, forming a highly stable thioether bond. This second reaction occurs optimally at a pH of 6.5-7.5. The resulting thioether linkage is a key feature of the SMCC crosslinker, rendering it "non-cleavable" under physiological conditions. This inherent stability is crucial for minimizing the premature release of the cytotoxic payload into systemic circulation, which in turn reduces off target toxicity and widens the therapeutic window of the ADC. The cytotoxic payload is only released after the ADC has bound to its target antigen on the cancer cell surface, been internalized, and trafficked to the lysosome, where the entire antibody is degraded, liberating the drug-linker-amino acid catabolite.

Synthesis of Peptide-Oligonucleotide Conjugates Using SMCC crosslinker
Peptide-oligonucleotide conjugates (POCs) possess unique properties that can be exploited in a wide range of applications from nanotechnology to drug delivery and antisense technology (Tung, 2000). Several synthetic protocols have been published that describe different approaches for making POC molecules. Fragment conjugation represents a convenient alternative to in-line synthesis. This unit describes the use of SMCC crosslinker to covalently link amine-modified DNA to cysteine-containing peptides. The protocol was adapted from a previously published method (Harrison and Balasubramanian, 1998), and requires the use of high purity (>95%) amine-modified DNA. A purification method using denaturing polyacrylamide gel electrophoresis (urea-PAGE) is described in Basic Protocol 1. The primary amine modification on the DNA is reacted with the NHS ester moiety of the SMCC reagent (Basic Protocol 2) to attach the bifunctional molecule to the DNA. The SMCC-modified DNA is then conjugated to the polypeptide by a Michael-type addition of the sulfhydryl group of the cysteine residue to the maleimide moiety of the SMCC crosslinker (Basic Protocol 3). Peptide-oligonucleotide conjugates can be purified using native-PAGE (Basic Protocol 4) or high performance liquid chromatography (HPLC).[1]
For modifications that occur at the 3’ terminus of the DNA strand, oligonucleotides can be synthesized from a universal linker or from a CPG column pre-charged with an amine moiety. The amine-modified DNA oligonucleotide must be purified prior to SMCC crosslinker conjugation in order to remove any primary amines that would otherwise compete with the DNA for the crosslinking reagent. Many companies provide oligonucleotide purification, however this additional service can be cost prohibitive for many labs. Fortunately, amine-modified oligonucleotides are easy to purify by denaturing urea-PAGE, and a standard procedure is given here. The fragment conjugation method presented here utilizes the heterobifunctional crosslinking reagent SMCC crosslinker. This method was originally reported by Harrison and Balasubramanian in 1997 to characterize the hybridization properties of POCs. The functionality of the SMCC molecule allows for a wide range of oligonucleotide and peptide sequences to be conjugated. This unit describes the synthesis and purification of POC molecules using the heterobifunctional crosslinking reagent succinimidyl-4-(N-maleimidomethyl)cyclohexane-1-carboxylate (SMCC crosslinker), which enables amine-modified oligonucleotides to become covalently linked to cysteine-modified polypeptides. This solution-based protocol consists of a two-step synthesis followed by a single purification step.
Establishment of SMCC crosslinker modified biochip
Lyme borreliosis (LB) is a multi-systemic disseminated disease, which is caused by Borrelia burgdorferi sensu lato by ticks of the species Ixodes. The typical symptom after infections often begins with erythema migrans (EM), and some patients would experience further symptoms, such as neurological, cardiac and arthritic abnormality if the patients have improper treatments. Neuroborreliosis (NB), including peripheral neuropathy and encephalomyelitis, may occur among some untreated patients months to years after tick-transmitted infection. In this study, we developed a SMCC crosslinker modified gold biochips for infectious antibody detection in NB. The general scheme used in the SMCC crosslinker based protocol to prepare maleimide-activated gold biochip and immobilization of proteins on the surface. Gold platform based microarray would be widely applied for various detections of bio-molecules such as nucleic acids, proteins, peptides and other small molecules. Surfaces with primary or secondary amino groups covalently bound were dedicated to promote the covalent immobilization of compounds containing reactive moieties such as amino, carboxyl or thiol groups via well-known homo- or hetero-bifunctional linkers, e.g. SMCC crosslinker or N-Hydroxysuccinimide. In our previous investigations, we constructed streptavidin-biotin based and N-hydroxysuccinimide (NHS) modified gold platforms for diagnosis of LB infections by detecting anti-flagellin antibody and anti-OspC 6-peptide antibody.[2]
It was demonstrated that modification of 16-amino-1-hexadecanethiol provided a long alkyl chain to reduce the steric hindrance effects of the subsequently probed bio-molecules (Wong et al., 2010). SMCC crosslinker is an amine-to-sulfhydryl crosslinker that contains NHS-ester and maleimide reactive group. As a hetero-bifunctional linker, SMCC was capable of offering an activated carboxyl group, covalently reacting with the sulfhydryl group on the proteins, and thereby exposing the active ends of these compounds to the liquid phase. We tested immobilization time courses and reaction temperatures of flagellin antigen on the SMCC crosslinker modified gold surfaces. Obviously, the interaction of flagellin antigen and antibody on the gold surfaces was time- and temperature- dependent. We proposed a SMCC crosslinker based chemical modification on gold biochip to detect serum immunogenicity of three B. burgdorferi antigens, flagellin, OspC and VlsE, in a panel of NB patients. The potential application of this protein biochip could be extended to test other molecules in the serum with high sensitivity and specificity. Due to the property of the biochip platform on detecting a larger panel of molecules in each individual serum, it would be possible to establish a combination biochip for detection of different molecules in one experimental outcome.
Synthesis of SMCC crosslinker as a Linker and Conjugating Trastuzumab
HER2 is overexpressed in 20–25% of breast cancer cases and is responsible for more aggressive tumor behavior. In order to increase the efficiency in cancer therapy, antibody–drug conjugates (ADCs) can be used that combine the antigen-targeting specificity of monoclonal antibodies with the cytotoxicity of chemotherapeutic drugs. All three components of ADCs affect the efficacy and toxicity of the drug. In this study, an ADC was synthesized by conjugating trastuzumab to maytansinol. SMCC crosslinker was synthesized in five different steps and compounds at each step were analyzed using infrared spectroscopy. The structure of the final product was confirmed using NMR. DM1 attached to SMCC crosslinker in chloroform solvent and was purified by recrystallization. Antibody purification was performed by chromatography and bound to SMCC-DM1. SMCC crosslinker was obtained with a 75.8% yield and melting point at 187–189°C. Conjugation of DM1 with SMCC occurred in acetonitrile. Antibodies with a concentration of 10 mg/mL and SMCC-DM1 with concentration of 20 mg/mL had the best results in terms of drug-bound antibody production. ADCs are a promising approach to the targeted therapy of cancer. One way to achieve safe and potent drugs is to optimize each component before synthesis, including the selection of the optimal specific antibody against the targeted antigen, the conjugation method, and the chemical properties of the linker.[3]
References
[1]Williams BA, Chaput JC. Synthesis of peptide-oligonucleotide conjugates using a heterobifunctional crosslinker. Curr Protoc Nucleic Acid Chem. 2010 Sep;Chapter 4:Unit4.41. doi: 10.1002/0471142700.nc0441s42. PMID: 20827717; PMCID: PMC2947322.
[2]Ye, Lei et al. “Establishment of N-succinimidyl 4-(maleimidomethyl) cyclohexanecarboxylate (SMCC) modified biochip enabling concurrent detection of serum infectious antibodies in neuroborreliosis.” Biosensors & bioelectronics vol. 78 (2016): 404-410. doi:10.1016/j.bios.2015.11.050
[3]Ahani, M., Salarian, M., Salehi, M., Jalili, N., Shafiee, S., Taheri, A., & Farahmand, L. (2024). Synthesis of Succinimidyl 4-(N-maleimidomethyl)-Cyclohexane-1-Carboxylate (SMCC) as a Linker and Conjugating Trastuzumab to Maytansinoid Derivative (DM1) Through SMCC as an Antibody-Drug Conjugate. Pharmaceutical Chemistry Journal, 57, 1384–1393.
Related articles And Qustion
Lastest Price from SMCC crosslinker manufacturers

US $0.00-0.00/KG2025-06-25
- CAS:
- 64987-85-5
- Min. Order:
- 1KG
- Purity:
- 99.8%
- Supply Ability:
- 100kg

US $10.00/kg2025-04-15
- CAS:
- 64987-85-5
- Min. Order:
- 1kg
- Purity:
- 99%
- Supply Ability:
- 20ton