Structural Properties and Biological Applications of Glycylglycine
Glycylglycine is a non-selective substrate for glycylglycine dipeptidase. Glycylglycine can cross cell membranes via passive diffusion and is hydrolyzed intracellularly to glycine, where it participates in energy metabolism and antioxidant processes. Glycylglycine promotes the proliferation of spermatogonial stem cells (SSCs), inhibits excessive activation of astrocytes, reduces the release of nitric oxide (NO), and upregulates the expression of neurotrophic factors (such as PDGFA, FGF2, and CNTF) to support neural myelin repair. Glycylglycine can be used in research on male reproductive biology (e.g., regulation of SSCs proliferation) and neurodegenerative diseases (e.g., neuroprotective mechanisms in multiple sclerosis).

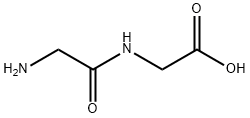
Figure1: Picture of Glycylglycine
Basic Introduction
Glycylglycine is the dipeptide of glycine, which makes it the simplest peptide, and it was first synthesized by Emil Fischer and Ernest Fourneau in 1901 through the boiling of 2,5-diketopiperazine (glycine anhydride) with hydrochloric acid, although other synthesis methods such as shaking with alkali have since been reported. Due to its low toxicity, glycylglycine is useful as a buffer for biological systems, exhibiting effective pH ranges between 2.5–3.8 and 7.5–8.9, though it is only moderately stable for storage once dissolved, and it is also employed in the synthesis of more complex peptides. Additionally, glycylglycine has been reported to be helpful in solubilizing recombinant proteins in E. coli, as improvements in protein solubility after cell lysis have been observed when using different concentrations of the compound. [1]
Structural Properties
In the structure of Glycylglycine, two glycine residues are closely linked through a peptide bond (-CO-NH-). The peptide bond is formed by a dehydration condensation reaction between the carboxyl group (-COOH) of one glycine and the amino group (-NH₂) of another glycine, a process that releases one molecule of water and connects the two amino acid residues to form a stable dipeptide structure. Its chemical structure is H₂NCH₂CONHCH₂COOH. Because the R group of glycine is a hydrogen atom, Glycylglycine possesses relatively high molecular flexibility, allowing it to adopt various conformations in solution. Meanwhile, the amino and carboxyl groups within the molecule exhibit different dissociation states under different pH conditions. In acidic solutions, the amino group binds hydrogen ions (H⁺), rendering the molecule positively charged, and under such conditions, Glycylglycine can interact with negatively charged molecules or groups through electrostatic attraction. In alkaline solutions, the carboxyl group loses hydrogen ions and becomes negatively charged, enabling Glycylglycine to interact with positively charged molecules or groups. These structural characteristics determine the unique chemical reactivity and biological functions of Glycylglycine; for instance, during protein folding, Glycylglycine may influence the three-dimensional structure of proteins through interactions with other amino acid residues.
Cell Viability Assay
In cell viability assays, Glycylglycine (10 μM; 5 days) significantly promotes cell proliferation in spermatogonial stem cell (SSC) cultures, increasing cell viability by approximately 50% without exerting any significant effects on apoptosis or the cell cycle. In contrast, Glycylglycine (0.1–1 mM; 24 hours) inhibits the growth of astrocytic U87 cells, while treatment with Glycylglycine (1 mM; 12–72 hours) shows no significant impact on DNA synthesis.
Biological Applications
Glycylglycine is a biochemical reagent used in biological research and medicine as a stabilizer for blood preservation and for aqueous injections of the protein drug cytochrome C. It serves as a substrate for the determination of glycylglycine dipeptidase and is also utilized in the synthesis of peptides. Furthermore, as a short-chain peptide, the interaction of Glycylglycine with transition metals plays a critical role in various fields such as bioengineering and medicinal chemistry.[2]
As an Enzyme Substrate
Glycylglycine serves as a specific substrate for glycylglycine dipeptidase. When Glycylglycine encounters this enzyme, the active site of the enzyme precisely recognizes and binds to Glycylglycine. This recognition process is based on specific intermolecular interactions, such as hydrogen bonds and van der Waals forces. Upon binding, the enzyme catalyzes the cleavage of the peptide bond, breaking Glycylglycine down into two glycine molecules. This enzymatic reaction plays a critical role in protein metabolism within living organisms, providing the foundation for protein synthesis, degradation, and the recycling of amino acids. For instance, during intracellular protein degradation, the glycine produced from the enzymatic hydrolysis of Glycylglycine can serve as a raw material for new protein synthesis, helping to maintain amino acid balance and normal physiological functions in the body.
Participation in Buffer Systems
Within the realm of biochemical research, Glycylglycine can function as a buffering substance. The amino group (-NH₂) and carboxyl group (-COOH) in its molecular structure possess unique chemical properties that allow them to undergo reversible reactions with hydrogen ions (H⁺) or hydroxide ions (OH⁻) in solution within a certain range. When the concentration of hydrogen ions in the solution increases, the amino group binds to hydrogen ions, thereby neutralizing acidity; when the concentration of hydroxide ions increases, the carboxyl group reacts with them, neutralizing alkalinity. In this way, Glycylglycine can regulate the pH of a solution and maintain acid-base balance in the system. In cell culture systems, a stable pH environment is crucial for normal cell growth, metabolism, and the maintenance of various physiological functions, and Glycylglycine can provide effective buffering in such systems to ensure the stability of the cellular environment. Additionally, in enzymatic reaction systems that require precise pH control, Glycylglycine can serve as an ideal buffer to ensure that enzyme activity remains at an optimal level. [3]
Interaction with Metal Ions
Glycylglycine can interact with transition metal ions. The nitrogen and oxygen atoms in its molecule possess lone pairs of electrons, allowing them to act as ligands and form coordination bonds with metal ions, thereby forming metal-Glycylglycine complexes. This interaction demonstrates significant value in multiple fields. In living organisms, metal-Glycylglycine complexes may influence the transport, storage, and metabolism of metal ions. For example, certain metal ions (such as iron and copper ions) require binding to specific ligands to remain stable in bodily fluids such as blood and be transported to the required tissues and cells, and Glycylglycine may participate in this process, playing an important role in maintaining metal ion homeostasis. In the field of materials science, the interaction between Glycylglycine and metal ions can be utilized to prepare materials with special properties. These materials may exhibit unique optical, electrical, or catalytic properties, offering potential applications in sensors, catalysts, nanomaterials, and other areas. For instance, by synthesizing metal-Glycylglycine nanocomposites, highly sensitive sensors for specific gases can be developed for environmental monitoring purposes.
Storage Conditions
From the perspective of stability, it is strongly recommended to store Glycylglycine at a low temperature of -20 °C. Under such low-temperature conditions, the thermal motion of molecules is significantly reduced, and the rate of chemical reactions is slowed, effectively minimizing decomposition, deterioration, and other issues that may arise from exposure to higher temperatures. If Glycylglycine requires frequent use over a short period, storage at 4 °C (refrigeration) is also acceptable; however, for long-term preservation, -20 °C remains the optimal choice. During storage, care must be taken to ensure proper sealing to prevent contact with air. Moisture and oxygen from the air may lead to hygroscopic absorption and oxidation of Glycylglycine, thereby affecting its quality and performance. Hygroscopic absorption may alter its actual concentration, while oxidation may cause changes in its molecular structure, resulting in the loss of its original biological activity and chemical properties. For example, Glycylglycine that has absorbed moisture may compromise the accuracy of experimental results when used due to concentration inaccuracies; oxidized Glycylglycine may fail to properly participate in relevant chemical reactions or biological processes. To further ensure its stability, a desiccant may be placed in the storage container to absorb any potential moisture.
Reference
[1] Abiogenic Syntheses of Lipoamino Acids and Lipopeptides and their Prebiotic Significance. Orig. Life Evol. Biosph. 45, 427 - 37, (2015).
[2] Recurrence of carbamoyl phosphate synthetase 1 (CPS1) deficiency in Turkish patients: characterization of a founder mutation by use of recombinant CPS1 from insect cells expression. Mol. Genet. Metab. 113, 267, 2014.
[3] Smith M E, Smith L B. Piperazine dihydrochloride and glycylglycine as non-toxic buffers in distilled water and in sea water[J]. The Biological Bulletin, 1949, 96:233-237.
You may like
Related articles And Qustion
Lastest Price from Glycylglycine manufacturers

US $500.00-500.00/kg2025-10-10
- CAS:
- 556-50-3
- Min. Order:
- 1kg
- Purity:
- ≥99.00%
- Supply Ability:
- 1000000

US $0.00-0.00/Kg/Drum2025-04-21
- CAS:
- 556-50-3
- Min. Order:
- 1KG
- Purity:
- 99%min
- Supply Ability:
- 1000kg/month