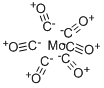Structure and properties of Molybdenum hexacarbonyl
Molybdenum hexacarbonyl, also referred to as molybdenum carbonyl, is a chemical compound represented by the formula Mo(CO)₆. As a colorless solid, molybdenum hexacarbonyl is notable for being a volatile and air-stable derivative of a metal in its zero oxidation state, similar to its chromium, tungsten, and seaborgium analogues.

Figure1: Picture of Molybdenum hexacarbonyl
Overview
Molybdenum hexacarbonyl (Mo(CO)₆) holds significant importance in organic synthesis. As a Group 6 element in the periodic table, molybdenum is a silvery-white metal that is tough yet malleable and softer than glass. It has an atomic mass of 95.94; the melting point of molybdenum hexacarbonyl is 2610 °C, and its boiling point is 5560 °C; its electron configuration is [Kr] 4d⁵5s¹. Molybdenum commonly exhibits six oxidation states, including –4, –2, 0, +2, +4, and +6. Molybdenum hexacarbonyl is a white, diamagnetic solid that is stable in air and moisture, odorless, and forms orthorhombic crystals with a density of 1.96 g·cm⁻³. Its structure is octahedral. The hydrophobic crystals sublime under vacuum at 142–146 °C and decompose without melting at 150 °C. It is slightly soluble in organic solvents such as THF, diglyme, and MeCN, but insoluble in water. A wide range of reactions can be catalyzed by molybdenum hexacarbonyl, including C–C bond-forming reactions, cyclization reactions, cleavage of heterocyclic rings, reductions, oxidation sequences, and the formation of heterocyclic rings. [1]
Structure and properties
Molybdenum hexacarbonyl adopts an octahedral geometry consisting of six rod‑like CO ligands radiating from the central Mo atom. A recurring minor debate in some chemical circles concerns the definition of an “organometallic” compound, where the term typically refers to compounds containing a metal directly bonded via an M–C bond to an organic fragment that in turn possesses a C–H bond. Like many metal carbonyls, molybdenum hexacarbonyl is generally prepared by reductive carbonylation, which involves the reduction of a metal halide under an atmosphere of carbon monoxide. As outlined in a 2023 survey of synthetic methods, the most cost‑effective routes for the synthesis of group 6 hexacarbonyls are based on the reduction of the corresponding metal chlorides—such as CrCl₃, MoCl₅, or WCl₆—using magnesium, zinc, or aluminum powders under carbon monoxide pressure.
Chemical Properties
Molybdenum hexacarbonyl is a widely used reagent in laboratory research. One or more of its CO ligands can be displaced by other ligands, and some of the resulting intermediates are well‑suited for further substitution reactions. For example, reacting [Mo(CO)₄(piperidine)₂] with triphenylphosphine yields cis‑ and trans‑[Mo(CO)₄(PPh₃)₂]. The compound [Mo(CO)₃(MeCN)₃] serves as a convenient source of “Mo(CO)₃” synthon; treatment with allyl chloride gives [MoCl(allyl)(CO)₂(MeCN)₂], while reactions with KTp and sodium cyclopentadienide produce the anions [MoTp(CO)₃]⁻ and [MoCp(CO)₃]⁻, respectively. These anions subsequently react with a variety of electrophiles. Another related source of Mo(CO)₃ is cycloheptatrienemolybdenum tricarbonyl.[2]
Occurrence
Molybdenum hexacarbonyl has been detected in landfills and sewage treatment plants, as the reducing and anaerobic conditions in such environments are conducive to its formation. [3]
Application
Molybdenum hexacarbonyl, along with derivatives such as [Mo(CO)₃(MeCN)₃], is employed as a catalyst or reagent in organic synthesis, including applications in alkyne metathesis and the Pauson–Khand reaction. In these processes, molybdenum hexacarbonyl can serve as a convenient source of carbon monoxide. Molybdenum hexacarbonyl is widely utilized in electron beam‑induced deposition techniques due to its easy vaporization and decomposition under an electron beam, thereby providing a readily available source of molybdenum atoms.
Handling and safety
It should be noted that Molybdenum hexacarbonyl is supplied as a white crystalline solid. It is stable at room temperature under nitrogen. One should avoid contact with the skin, eyes, and vapor inhalation. All the reactions should be carried out in an efficient fume hood with proper protective equipment due to the involvement of CO generation under the reaction conditions. [1]
Reference
[1] Kotha S, Gaikwad V, Chaurasia U N. Application of molybdenum hexacarbonyl in organic synthesis[J]. Tetrahedron, 2024, 167: 134264.
[2] Bruno S M, Valente A A, Gon?alves I S, et al. Group 6 carbonyl complexes of N, O, P-ligands as precursors of high-valent metal-oxo catalysts for olefin epoxidation[J]. Coordination Chemistry Reviews, 2023, 478: 214983.
[3] Feldmann J. Determination of Ni (CO) 4, Fe (CO) 5, Mo (CO) 6, and W (CO) 6 in sewage gas by using cryotrapping gas chromatography inductively coupled plasma mass spectrometry[J]. Journal of Environmental Monitoring, 1999, 1: 33-37.