Study on the Clinical Pharmacological Effects of Epalrestat
Introduction
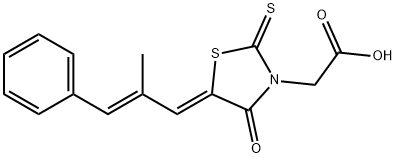
Epalrestat ((5-[(1Z,2E)-2-methyl-3-phenylpropenylidene]-4-oxo-2-thioxo-3-thiazolidineacetic acid);fig. 1) is a phenylpropenylidenethiazolidine carboxylic acid, one of several chemically diverse carboxylic acid derivatives which have been found to inhibit aldose reductase, and thus has potential for ameliorating late-onset complications of diabetes mellitus. Epalrestat was reformulated during its development, resulting in significantly improved absorption following oral administration. Thus, the currently recommended dosage of 50mg 3 times daily before meals is considerably less than that used in earlier clinical trials. Epalrestat is a carboxylic acid derivative which inhibits aldose reductase, an enzyme of thesorbitol (polyol) pathway. Under hyperglycaemic conditions epalrestat reduces intracellular sorbitol accumulation, which has been implicated in the pathogenesis of late-onset complications of diabetes mellitus. [1]

Bioequivalence Study of Epalrestat for Healthy Chinese Subjects
Epalrestat is a reversible noncompetitive inhibitor of aldose reductase with selective inhibition of aldose reductase. It can inhibit the accumulation of sorbitol in red blood cells in patients with diabetic peripheral neuropathy and can improve patients' conscious symptoms and neurological dysfunction. This study was designed to evaluate the bioequivalence in healthy Chinese subjects of a new test formulation and reference formulation of oral epalrestat (50 mg) in the fasting state. The study was performed with 44 healthy Chinese subjects according to a randomized 2-way crossover design. The main pharmacokinetic parameters of test formulation and reference formulation as follows: 4793 and 4781 ng/mL for maximum plasma concentration, 8556 and 8431 ng h/mL for area under the plasma concentration-time curve extrapolated to infinity. The test formulation of epalrestat was bioequivalent to the reference formulation. The bioequivalence study of epalrestat in healthy Chinese subjects suggests that the test and reference formulations have similar pharmacokinetics and both formulations are well tolerated in the dose range studied in healthy Chinese subjects. All these findings provided valuable pharmacokinetic knowledge for further clinical development.[2]
Alpha lipoic acid combined with epalrestat: a therapeutic option for patients with diabetic peripheral neuropathy
Alpha lipoic acid (ALA), a type of antioxidant, is used in combination with epalrestat in the treatment of diabetic peripheral neuropathy (DPN). However, whether combined treatment is superior to epalrestat monotherapy is controversial. Wang et al. conducted a systematic search of PubMed, Cochrane Library and Chinese databases to identify all randomized controlled trials (RCTs) up to October 31, 2017. Data were extracted to evaluate methodological quality and analyzed using Review Manager 5.3.0 software. Twelve studies were included. Compared to epalrestat monotherapy, ALA 600 mg/d once a day (qd) combined with epalrestat 50 mg three times a day (tid) augmented the total effectiveness rate (14 days - risk ratio [RR]: 1.40, 95% CI: 1.16-1.69, P=0.0005; 28 days - RR: 1.48, 95% CI: 1.27-1.72, P<0.00001); at the same, it could improve the median motor nerve conduction velocity (MNCV) and sensory nerve conduction velocity (SNCV), peroneal MNCV, and SNCV after 14, 21, and 28 days of treatment and could reduce the Toronto Clinical Scoring System (TCSS) (weighted mean difference [WMD]: -1.60, 95% CI: (-2.91, -0.29), P=0.02) and Total Symptom Score (TSS) (WMD: -0.93, 95% CI: -1.27, -0.60, P<0.00001) after 21 days of treatment. The treatment strategy of ALA 300 mg/d qd combined with epalrestat 50 mg tid had the same effects in regard to the total effectiveness rate (RR: 1.37, 95% CI: 1.18-1.59, P<0.0001), median MNCV (WMD: 6.12, 95% CI: 5.04, 7.20, P=0.00001), median SNCV (WMD: 6.70, 95% CI: 5.75, 7.65, P=0.00001), peroneal MNCV (WMD: 6.68, 95% CI: 5.82, 7.55, P=0.00001), and peroneal SNCV (WMD: 4.27, 95% CI: 3.34, 5.20, P=0.00001) after 28 days of treatment.[3]
Targeting AKR1B10 by Drug Repurposing with Epalrestat Overcomes Chemoresistance in NSCLC
Systemic treatments given to patients with non-small cell lung cancer (NSCLC) are often ineffective due to drug resistance. In the present study, we investigated patient-derived tumor organoids (PDTO) and matched tumor tissues from surgically treated patients with NSCLC to identify drug repurposing targets to overcome resistance toward standard-of-care platinum-based doublet chemotherapy. PDTOs were established from 10 prospectively enrolled patients with non-metastatic NSCLC from resected tumors. PDTOs were compared with matched tumor tissues by histopathology/immunohistochemistry, whole exome sequencing, and transcriptome sequencing. PDTO growths and drug responses were determined by measuring 3D tumoroid volumes, cell viability, and proliferation/apoptosis. Differential gene expression analysis identified drug-repurposing targets. Validations were performed with internal/external data sets of patients with NSCLC. NSCLC cell lines were used for aldo-keto reductase 1B10 (AKR1B10) knockdown studies and xenograft models to determine the intratumoral bioavailability of epalrestat.
PDTOs retained histomorphology and pathological biomarker expression, mutational/transcriptomic signatures, and cellular heterogeneity of the matched tumor tissues. Five (50%) PDTOs were chemoresistant toward carboplatin/paclitaxel. Chemoresistant PDTOs and matched tumor tissues demonstrated overexpression of AKR1B10. Epalrestat, an orally available AKR1B10 inhibitor in clinical use for diabetic polyneuropathy, was repurposed to overcome chemoresistance of PDTOs. In vivo efficacy of epalrestat to overcome drug resistance corresponded to intratumoral epalrestat levels. PDTOs are efficient preclinical models recapitulating the tumor characteristics and are suitable for drug testing. AKR1B10 can be targeted by repurposing epalrestat to overcome chemoresistance in NSCLC. Epalrestat has the potential to advance to clinical trials in patients with drug-resistant NSCLC due to favorable toxicity, pharmacological profile, and bioavailability.[4]
References
[1] Steele JW, Faulds D, Goa KL. Epalrestat. A review of its pharmacology, and therapeutic potential in late-onset complications of diabetes mellitus. Drugs Aging. 1993;3(6):532-555. doi:10.2165/00002512-199303060-00007
[2] Yang D, Wang X, Duan Y, et al. Bioequivalence Study of Epalrestat for Healthy Chinese Subjects. Clin Pharmacol Drug Dev. 2024;13(5):485-490. doi:10.1002/cpdd.1347
[3] Wang X, Lin H, Xu S, Jin Y, Zhang R. Alpha lipoic acid combined with epalrestat: a therapeutic option for patients with diabetic peripheral neuropathy. Drug Des Devel Ther. 2018;12:2827-2840. Published 2018 Sep 7. doi:10.2147/DDDT.S168878
[4] Suvilesh KN, Manjunath Y, Nussbaum YI, et al. Targeting AKR1B10 by Drug Repurposing with Epalrestat Overcomes Chemoresistance in Non-Small Cell Lung Cancer Patient-Derived Tumor Organoids. Clin Cancer Res. 2024;30(17):3855-3867. doi:10.1158/1078-0432.CCR-23-3980
You may like
Related articles And Qustion
See also
Lastest Price from Epalrestat manufacturers

US $0.00/Kg/Bag2025-04-21
- CAS:
- 82159-09-9
- Min. Order:
- 1KG
- Purity:
- 99%min
- Supply Ability:
- 100KGS

US $10.00/KG2025-03-13
- CAS:
- 82159-09-9
- Min. Order:
- 1KG
- Purity:
- 99%
- Supply Ability:
- 10 mt