Synthesis and application of Norandrostenedione
Introduction
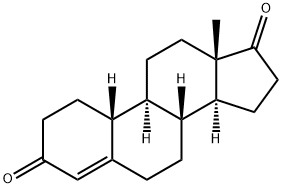
One of the most frequently misused steroid precursors (prohormones) is norandrostenedione (4-estrene-3,17-dione, NOR), which is, after oral administration, readily metabolised to nortestosterone, also known as nandrolone (durabolin).Together with dehydroepiandrosterone and androstenedione, norandrostenedione is frequently designated as over-the-counter steroid. Norandrostenedione refers to two steroid isomers that are marketed as dietary supplements and mainly used by body builders. The difference between the two norandrostenedione isomers is in the position of the double bond in the cyclopentanoperhydrophenanthrene ring structure. Norandrostenedione refers to the absence of a 19 methyl group on the steroid ring structure. Norandrostenedione is synthesized in the adrenal gland and gonads from androstenedione. It is metabolized by the aromatase complex to estrone.The △4, as well as △5, norandrostenedione is metabolized by the enzyme 17 beta-hydroxy steroid dehydrogenase to nortestosterone, also known as nandrolone in both men and women (Fig. 1).[1]

Synthesis of Norandrostenedione
Method 1: starting from the δ-Lactone,norandrostenedione was synthesized by Grignard reaction, oxidation, ring closure, reduction, deprotection, and reclosed route.The total yield was 59.6%.[2]
Method 2:Norandrostenedione was synthesized in vitro from dehydroepiandrosterone by explants of equine full-term placenta. The synthesis of norandrostenedione was inhibited by two specific aromatase inhibitors, 4-hydroxyandrostenedione and fadrozole.[3]
Method 3:The origin and biosynthesis of 4-oestrene-3,17-dione (norandrostenedione), a major steroid in porcine ovarian follicular fluid, was investigated by culturing granulosa cells from 4-6 mm follicles of prepubertal gilts with radiolabelled androstenedione and hydroxyandrostenedione. Steroid metabolites were purified by solvent extraction and lipophilic column chromatography, and analysed by C18 reverse-phase high-performance liquid chromatography. 19-Hydroxyandrostenedione, norandrostenedione and oestradiol-17 beta were obtained as major metabolites from androstenedione, while norandrostenedione and oestradiol-17 beta were the major products from 19-hydroxyandrostenedione. Serum alone or serum plus FSH significantly enhanced formation of norandrostenedione and oestradiol-17 beta from each substrate, compared with controls. Micromolar concentrations (1 mumol/l) of 4-hydroxyandrostenedione, an aromatase inhibitor, significantly reduced formation of norandrostenedione and oestradiol-17 beta by granulosa cells cultured with serum and FSH. Formation of norandrostenedione and oestradiol-17 beta from androstenedione and 19-hydroxyandrostenedione was also significantly inhibited by aminoglutethimide phosphate, a cytochrome P-450 inhibitor known to block the conversion of androstenedione to oestrogens. Ketoconazole, an inhibitor of the cytochrome P-450 dependent 17,20-lysase, blocked formation of 19-norandrostenedione and oestradiol-17 beta only at millimolar concentrations. These results suggest that 19-norsteroid and oestrogen formation from C19 aromatizable androgens may share a common or overlapping pathway, and imply that 19-norsteroid and oestrogen synthesis is mediated by cytochrome P-450 dependent enzymes.[4]
Anabolic and androgenic activity of norandrostenedione after oral and subcutaneous administration
One of the most frequently misused steroid precursors (prohormones) is norandrostenedione (estr-4-ene-3,17-dione, NOR). Recently Parr et al. have show that norandrostenedione stimulates skeletal muscle growth after s.c. administration in a highly selective manner but exhibits only weak androgenic activity in rats. Because most abusers take norandrostenedione orally, the aim of this study was to compare the anabolic and androgenic potency of NOR between s.c. and oral application. Orchiectomised rats were treated with norandrostenedione either s.c. (1 mg/kg BW/day) or orally (0.1, 1 and 10 mg/kg BW/day). The tissue weights of the levator ani, the seminal vesicle and the prostate were analysed to determine the anabolic and androgenic activity. Heart and liver wet weights were examined to identify side effects. Serum concentrations of NOR and its metabolite nandrolone (NT) were determined. GCMC analysis revealed that free and glucuronidated NOR and NT were detectable in the serum after oral and s.c. administration and that norandrostenedione was converted to NT in comparable amounts independent of the route of administration. In agreement to our previous study s.c. application of NOR stimulates skeletal muscle growth but has only weak androgenic effects. In contrast, after oral administration of norandrostenedione neither stimulation of the prostate nor the levator ani could be observed in the doses administered in this study. Interestingly, and in contrast to s.c. treatment, oral administration of norandrostenedione resulted in a dose-dependent decrease of body weight. In summary, oral administration of norandrostenedione, at least in the rat, seems to be a very ineffective strategy for stimulating skeletal muscle mass increases but may be associated with side effects.[5]
Hydroxylation of norandrostenedione
The activity of purified bovine adrenocortical P-450(11)beta on the C18-steroid, 4-estrene-3,17-dione (norandrostenedione), is described. The major steroid products were separated by HPLC and identified by GC-MS, and 1H- and 13C-NMR as 11 beta-, 18- and 6 beta-hydroxylated derivatives of norandrostenedione. The turnover numbers of the 11 beta-, 18- and 6 beta-hydroxylase reactions were 45, 7.5 and 1.9 (mol/min/mol of P-450(11)beta), respectively, with a common Km of 44 microM. All of these activities required the presence of the electron donating system consisting of NADPH, adrenal ferredoxin (adrenodoxin) and its reductase. These findings provide additional insights into the versatile catalytic roles of P-450(11)beta in the adrenal cortex, in which it may act on C18-19-nor-steroids in addition to its known activities on C21- and C19-steroids.[6]
References
[1] Diel P, Friedel A, Geyer H, et al. The prohormone 19-norandrostenedione displays selective androgen receptor modulator (SARM) like properties after subcutaneous administration. Toxicol Lett. 2008;177(3):198-204. doi:10.1016/j.toxlet.2008.01.014
[2] Cui Zk. A Novel Synthetic Route For Estra-4,9-diene-3,17-dione and 19-nor-4-androstenedione[D].Zhejiang Sci-Tech University, 2019.DOI:10.27786/d.cnki.gzjlg.2019.000093.
[3] Moslemi S, Silberzahn P, Gaillard JL. The synthesis of 19-norandrostenedione from dehydroepiandrosterone in equine placenta is inhibited by aromatase inhibitors 4-hydroxyandrostenedione and fadrozole. Comp Biochem Physiol B Biochem Mol Biol. 1995;112(4):613-618. doi:10.1016/0305-0491(95)00117-4
[4] Khalil MW, Morley P, Glasier MA, Armstrong DT, Lang T. Formation of 4-oestrene-3,17-dione (19-norandrostenedione) by porcine granulosa cells in vitro is inhibited by the aromatase inhibitor 4-hydroxyandrostenedione and the cytochrome P-450 inhibitors aminoglutethimide phosphate and ketoconazole. J Endocrinol. 1989;120(2):251-260. doi:10.1677/joe.0.1200251
[5] Parr MK, Laudenbach-Leschowsky U, Höfer N, Schänzer W, Diel P. Anabolic and androgenic activity of 19-norandrostenedione after oral and subcutaneous administration--analysis of side effects and metabolism. Toxicol Lett. 2009;188(2):137-141. doi:10.1016/j.toxlet.2009.03.024
[6] Suhara K, Yamamoto M, Katagiri M. Hydroxylation of 19-norandrostenedione by adrenal cortex mitochondrial P-450(11)beta. J Steroid Biochem. 1990;36(4):361-367. doi:10.1016/0022-4731(90)90230-pYou may like
Lastest Price from Norandrostenedione manufacturers

US $5.00-0.50/KG2025-05-30
- CAS:
- 734-32-7
- Min. Order:
- 1KG
- Purity:
- 99% hplc
- Supply Ability:
- 500TONS

US $0.00/kg2025-03-18
- CAS:
- 734-32-7
- Min. Order:
- 1kg
- Purity:
- 0.99
- Supply Ability:
- 10T