Synthesis and Application research of N-Ethylcarbazole
Introduction
Liquid Organic Hydrogen Carriers (LOHCs) hydrogen storage has the advantages of high hydrogen storage density, good safety, high reversibility and convenient transportation, which makes it a promising hydrogen storage technology for application.N-ethylcarbazole (NEC;Figure 1 ), which has a hydrogen storage capacity of 5.79 wt% and a unique nitrogen heterocyclic ring in its structure, significantly reduces the enthalpy of dehydrogenation and the temperature of the dehydrogenation reaction and facilitates the release of H2, and is considered as one of the ideal LOHCs. [1] As a new liquid organic hydrogen storage carrier, N-ethylcarbazole can be completely hydrogenated to perhydro N-ethylarbazole, which can be stored and transported in the same way as common oil products used. Because of the high mass density and the lower dehydrogenation temperature compared with the traditional materials, N-ethylcarbazole has been considered as one of the most promising hydrogen carriers.[2]

Ultrasonic Synthesis of Hydrogen Storage Material N-ethylcarbazole
Using carbazole and diethyl sulfate as raw materials, hydrogen storage N-ethylcarbazole was prepared by ultrasonic reaction, and pure N-ethylcarbazole was obtained by further treatment of the reaction solution.The effects of solvent, reaction temperature and reaction time on the yield of N-ethylcarbazole were investigated.The results showed that the best solvent was acetone, the best reaction temperature was 40 °C, ultrasonic power was 100 W,irradiation frequency was 40 kHz, and the best reaction time was 1 min.The structure of the product was characterized by FTIR and 1H-NMR. The liquid chromatographic purity of N-ethylcarbazole was >99.5%, the gas chromatographic purity was >99.5%, the melting point was 68°C, the total yield was 99%, and the synthesized product could be used as hydrogen storage material.[3]
Catalytic performance for hydrogen storage of N-ethylcarbazole
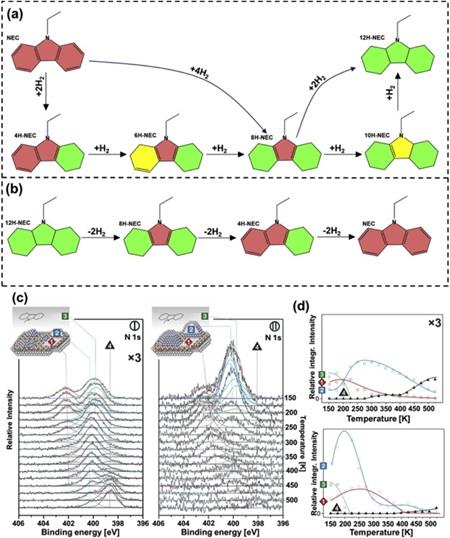
N-ethylcarbazole (NEC) is an ideal liquid organic hydrogen storage carrier. The development of efficient hydrogen storage catalysts can promote the large-scale application of this process. In this paper, SBA-15 supported Ru nanocatalysts (Ru/S15-SU) were synthesized by strong electrostatic adsorption (SEA)-ultrasonic in situ reduction method (UR). Ru/S15-SU was characterized by N2 adsorption-desorption, TEM, H2 temperature program reduction, FT-IR, XRD, and XPS analysis measures. The results showed that ultrafine Ru NPs were evenly distributed on the surface of SBA-15, and ultrasonic in situ reduction not only reduced Ru3+ to Ru0, but also produced a coordination effect between Ru and O, enhancing the interaction between Ru NPs and the carrier. Ru/S15-SU exhibited excellent catalytic performance in the hydrogenation reaction of NEC, and the hydrogen storage efficiency reached 99.31% at 130°C and 6 MPa H2 pressure, which is superior to that of commercial 5wt%Ru/Al2O3. The excellent catalytic hydrogenation performance can be attributed to the selective anchoring of ruthenium ions on the surface of SBA-15 via electrostatic adsorption, preventing the aggregation of Ru NPs and enhancing the interaction between SBA-15 and Ru NPs by ultrasonic in situ reduction. Ru/S15-SU had a lower NEC hydrogenation apparent activated energy (Ea) of 68.45 kJ/mol than 5wt%Ru/Al2O3 catalyst. This method provides a new approach for the green preparation of nanocatalysts without using any chemical reducing agents.[1]
Photochemically activated polymerizations of N-ethylcarbazole and 9-phenylcarbazole
The polymerization of photoexcited N-ethylcarbazole (N-EC) in the presence of an electron acceptor begins with an electron transfer (ET) step to generate a radical cation of N-EC (N-EC˙+). Here, the production of N-EC˙+ is studied on picosecond to nanosecond timescales after N-EC photoexcitation at a wavelength λex = 345 nm using transient electronic and vibrational absorption spectroscopy. The kinetics and mechanisms of ET to diphenyliodonium hexafluorophosphate (Ph2I+PF6-) or para-alkylated variants are examined in dichloromethane (DCM) and acetonitrile (ACN) solutions. The generation of N-EC˙+ is well described by a diffusional kinetic model based on Smoluchowski theory: with Ph2I+PF6-, the derived bimolecular rate coefficient for ET is kET = (1.8 ± 0.5) × 1010 M-1 s-1 in DCM, which is consistent with diffusion-limited kinetics. This ET occurs from the first excited singlet (S1) state of N-ethylcarbazole, in competition with intersystem crossing to populate the triplet (T1) state, from which ET may also arise. A faster component of the ET reaction suggests pre-formation of a ground-state complex between N-EC and the electron acceptor. In ACN, the contribution from pre-reaction complexes is smaller, and the derived ET rate coefficient is kET = (1.0 ± 0.3) × 1010 M-1 s-1. Corresponding measurements for solutions of photoexcited 9-phenylcarbazole (9-PC) and Ph2I+PF6- give kET = (5 ± 1) × 109 M-1 s-1 in DCM. Structural modifications of the electron acceptor to increase its steric bulk reduce the magnitude of kET: methyl and t-butyl additions to the para positions of the phenyl rings (para Me2Ph2I+PF6- and t-butyl-Ph2I+PF6-) respectively give kET = (1.2 ± 0.3) × 1010 M-1 s-1 and kET = (5.4 ± 1.5) × 109 M-1 s-1 for reaction with photoexcited N-ethylcarbazole in DCM. These reductions in kET are attributed to slower rates of diffusion or to steric constraints in the ET reaction.[4]
A N-Ethylcarbazole-Terminated Spiro-Type Hole-Transporting Material Preparation
The development of stable and efficient hole-transporting materials (HTMs) is critical for the commercialization of perovskite solar cells (PSCs). Herein, a novel spiro-type HTM was designed and synthesized where N-ethylcarbazole-terminated groups fully substituted the methoxy group of spiro-OMeTAD, named spiro-carbazole. The developed molecule exhibited a lower highest occupied molecular orbital level, higher hole mobility, and extremely high glass transition temperature (Tg=196 °C) compared with spiro-OMeTAD. PSCs with the developed molecule exhibited a champion power conversion efficiency (PCE) of 22.01 %, which surpassed traditional spiro-OMeTAD (21.12 %). Importantly, the spiro-carbazole-based device had dramatically better thermal, humid, and long-term stability than spiro-OMeTAD.[5]
Photoinduced Step-Growth Polymerization of N-Ethylcarbazole
A novel method for photochemical step-growth synthesis of poly( N-ethylcarbazole) (PEC) via consecutive diphenyl iodonium hexafluorophospate (Ph2I+PF6-) mediated electron transfer and coupling reactions is reported. The photoinduced electron transfer reaction of the excited N-ethylcarbazole (EC) in the presence of Ph2I+PF6- as the oxidizing salt proceeded efficiently giving N-ethylcarbazole radical cations (EC+•). Subsequently, the protons released concomitantly with coupling of two N-ethylcarbazole radical cations. The successive reactions involving excitation, electron transfer, proton release, and coupling lead to the formation of PEC. The electrochemical properties and surface morphology of the thin films of the formed polymers before and after dedoping were investigated by cyclic voltammetry, differential pulse voltammetry, and atomic force microscopy techniques, respectively.[6]
References
[1] Liu Ty.Study on Hydrogen Storage Performance of N-Ethylcarbazole Catalyzed by SBA-15-Supported Ruthenium-Based Catalysts[D].Heilongjiang University,2024.DOI:10.27123/d.cnki.ghlju.2024.000104.
[2] Chen B. Study on the hydrogen storage performance of N-ethylcarbazoleover Ni-based catalysts[D].Tianjin University of Science and Technology,2022.DOI:10.27359/d.cnki.gtqgu.2022.000988.
[3] XV Hr,et al.,Ultrasonic Synthesis of Hydrogen Storage Material N-ethylcarbazole[J].Contemporary Chemical Industry,2022,51(12):2874-2877.DOI:10.13840/j.cnki.cn21-1457/tq.2022.12.025.
[4] Thornton GL, Phelps R, Orr-Ewing AJ. Transient absorption spectroscopy of the electron transfer step in the photochemically activated polymerizations of N-ethylcarbazole and 9-phenylcarbazole. Phys Chem Chem Phys. 2021;23(34):18378-18392. doi:10.1039/d1cp03137f
[5] Han M, Liang Y, Chen J, et al. A N-Ethylcarbazole-Terminated Spiro-Type Hole-Transporting Material for Efficient and Stable Perovskite Solar Cells. ChemSusChem. 2022;15(20):e202201485. doi:10.1002/cssc.202201485
[6] Sari E, Yilmaz G, Koyuncu S, Yagci Y. Photoinduced Step-Growth Polymerization of N-Ethylcarbazole. J Am Chem Soc. 2018;140(40):12728-12731. doi:10.1021/jacs.8b08668
You may like
Related articles And Qustion
Lastest Price from N-Ethylcarbazole manufacturers
US $1.10-9.90/kg2025-08-15
- CAS:
- 86-28-2
- Min. Order:
- 100kg
- Purity:
- 99%min
- Supply Ability:
- 100kg

US $0.00/KG2025-04-21
- CAS:
- 86-28-2
- Min. Order:
- 1KG
- Purity:
- 98%min
- Supply Ability:
- 30tons/month