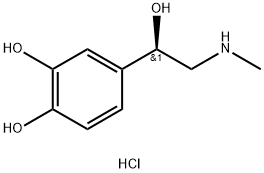
Synthesis and Approval Status of Epinephrine Hydrochloride
Epinephrine Hydrochloride typically exists as a white or off-white crystalline powder, which is prone to oxidative degradation upon exposure to air or light and exhibits instability in neutral or alkaline aqueous solutions, with only slight solubility in water. In its chemical structure, Epinephrine Hydrochloride contains a chiral carbon, giving rise to both levo- and dextrorotatory isomers, where the dextrorotatory form demonstrates stronger biological activity, although the racemic mixture is commonly used in practice. It is primarily metabolized in the liver and kidneys into norepinephrine and vanillylmandelic acid—metabolites that retain vasoconstrictive effects, albeit weaker than those of epinephrine—and has a half-life of approximately 2 to 3 minutes.
Synthesis
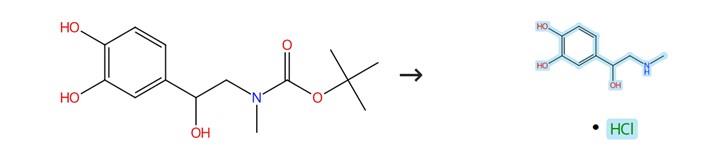
Figure1: Synthesis of Epinephrine Hydrochloride
The N-Boc-protected amines (1.0 mmol) were dissolved in methanol (20 mL), followed by the addition of trimethylsilyl chloride (TMSCl, 217 mg, 2.0 mmol). The resulting mixture was maintained under reflux conditions for 3 hours. After completion of the reaction, the solvent was carefully removed under reduced pressure, which allowed for the isolation of the corresponding amine hydrochloride derivative Epinephrine Hydrochloride in quantitative yield. [1]
Availability of Epinephrine Hydrochloride
To ascertain the availability of Epinephrine Hydrochloride (or epinephrine without a specified salt form) in the United States and globally, researchers conducted a systematic search of the national medicine registers across 13 countries and regions. Utilizing resources such as the World Health Organization, the European Medicines Agency (EMA), and globalEDGE, regulatory authorities outside the United States were identified. Inclusion criteria for these non-U.S. regulatory databases required that they be freely accessible, searchable, and capable of returning results in English, while also providing key information—including product trade name, active ingredient, strength, dosage form, route of administration, and approval status—in a structured and usable format. Based on these criteria, the following 13 countries and regions were included in the search: the United States, Canada, the European Union (EU), the United Kingdom (UK), Ireland, Belgium, Latvia, Australia, New Zealand, Saudi Arabia, Abu Dhabi, Hong Kong, and Namibia. To account for discrepancies in drug authorization between EU-level approvals and individual member states, both the EMA database and the national registers of selected EU countries (Ireland, the UK, Belgium, and Latvia) were examined in parallel. [2]
Approval Status
Epinephrine Hydrochloride was nominated for inclusion on the 503B Bulks List due to its broad therapeutic applicability across multiple medical conditions, administered through both single-agent and multi-agent preparation forms. As an FDA-approved product, epinephrine is commercially available in various solution formulations for intraocular, intramuscular (IM), intravenous (IV), and subcutaneous (SC) administration, and it is also supplied in a fixed-dose combination with bupivacaine hydrochloride for injectable use. Specifically, Epinephrine Hydrochloride has been formulated in combination with lidocaine as a 2% topical solution and with prilocaine as a 1–2% injectable preparation; however, these combination products have been discontinued for reasons unrelated to safety or efficacy. Furthermore, in regions including Abu Dhabi, Australia, Belgium, Canada, Hong Kong, Ireland, Latvia, Namibia, New Zealand, Saudi Arabia, and the United Kingdom, epinephrine remains accessible in the nominated dosage forms and corresponding routes of administration. [2]
Application
Epinephrine Hydrochloride is primarily used to treat severe dyspnea caused by bronchospasm, providing rapid relief for anaphylactic shock induced by medications or allergic reactions, and it can also be employed to prolong the duration of infiltration anesthesia. Furthermore, Epinephrine Hydrochloride serves as a key resuscitation agent in cardiopulmonary resuscitation for cardiac arrest resulting from various etiologies. In research settings, it has been applied in studies such as inducing renalase expression in human renal proximal tubular epithelial cells, examining its physiological effects in cattle through heart rate and ocular temperature measurements, and functioning as a supplement in culture media to stimulate endothelial progenitor cells.
Reference
[1] Bernini, Roberta; et al, A new and efficient route for the synthesis of naturally occurring catecholamines, Synthesis 2009, 22, 3838-3842.
[2] Yuen M V, Gianturco S L, Pavlech L L, et al. Epinephrine hydrochloride: Summary Report[J]. 2021.
You may like
See also
Lastest Price from Epinephrine Hydrochloride manufacturers

US $0.00-0.00/Kg2025-06-06
- CAS:
- 55-31-2
- Min. Order:
- 1Kg
- Purity:
- 99%
- Supply Ability:
- 50KG

US $5.00-0.50/KG2025-05-07
- CAS:
- 55-31-2
- Min. Order:
- 1KG
- Purity:
- 99% hplc
- Supply Ability:
- 500TONS