Synthesis and Chemical Applications of 2'-Chloroacetophenone
2'-Chloroacetophenone is a halogenated acetophenone derivative that appears as a colorless to pale yellow liquid under ambient temperature and pressure, exhibiting physicochemical properties similar to those of acetophenone. It shows limited solubility in water but is miscible with most organic solvents. Primarily used as an organic synthesis intermediate and a raw material in pharmaceutical manufacturing, 2'-Chloroacetophenone can be prepared from o-nitroacetophenone via a denitro-chlorination reaction. Studies have reported its application in the synthesis of the drug molecule Clorprenaline Hydrochloride (Chuantong).
Synthesis
Method 1
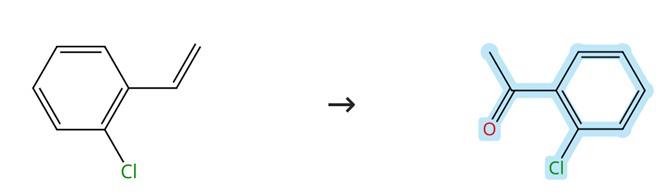
Figure1: Synthesis of 2'-Chloroacetophenone
To carry out the reaction, charge a 10 mL reaction tube equipped with a magnetic stir bar with the reactant (1.0 mmol), Pd(OAc)₂ (0.01 mmol), 1-(6-phenylpyridin-2-yl)-1H-indazole (0.05 mmol), K₂S₂O₈ (3.0 mmol), H₂O (0.3 mL), and EtOH (3.0 mL), and stir the mixture at 60°C for 12 hours. After completion of the reaction, allow the mixture to cool to room temperature, then dilute it with dichloromethane. Filter the resulting mixture through a pad of Celite to remove solid residues, and subsequently concentrate the filtrate under reduced pressure to remove the solvent. Finally, purify the crude residue by silica gel column chromatography using a gradient of petroleum ether and ethyl acetate (PET:EtOAc = 25:1) as the eluent to obtain the desired product 2'-Chloroacetophenone. [1]
Method 2

Figure2: Synthesis of 2'-Chloroacetophenone
In a 25 mL Schlenk tube equipped with a magnetic stir bar, combine the alcohol substrate (0.3 mmol, 1.0 equiv.), FeBr₃ (8.9 mg, 10 mol%), and acetonitrile (3 mL). Seal the tube with a rubber stopper and attach an air balloon to the sidearm. Place the assembled setup in a photoreactor equipped with a 10 W blue LED and stir the reaction mixture at 500 rpm for 4 hours. After irradiation, transfer the mixture to a separatory funnel, add 5 mL of water, and extract the aqueous mixture with ethyl acetate (3 × 10 mL). Combine the organic layers and wash them with saturated NaCl solution, then dry the organic phase over anhydrous Na₂SO₄. Filter the dried mixture to remove the desiccant, and concentrate the filtrate under reduced pressure to obtain the crude product. Finally, purify the crude material by column chromatography on silica gel using a gradient elution of petroleum ether/ethyl acetate (PE/EA = 100/1 to 10/1). Evaporate the solvent from the purified fractions under reduced pressure to yield the target compound, 2'-Chloroacetophenone. [2]
Chemical Applications
Synthesis of Clorprenaline Hydrochloride
Chuantong (Clorprenaline Hydrochloride) is an adrenergic β-receptor agonist that exhibits certain efficacy in relieving asthma, cough, and expectoration. It is a relatively new chemically synthesized drug developed domestically in recent years for the treatment of chronic bronchitis. 2'-Chloroacetophenone serves as a crucial intermediate in the synthesis of Chuantong. Typically, it is synthesized starting from o-chlorobenzoic acid via the Walker and Hauser ketone synthesis method. Although this method offers advantages such as stable process conditions and relatively high yield, 2'-Chloroacetophenone production via this route requires a large amount of raw materials.
Synthesis of o-Chlorostyrene
o-Chlorostyrene is prepared through phase-transfer catalytic reduction, dehydration, and distillation processes using o-chloroacetophenone as the starting material. The overall process yield is 74%, with a purity exceeding 99%. 2'-Chloroacetophenone is reduced in the presence of benzyltriethylammonium chloride as the phase-transfer catalyst (catalyst dosage: 2% by mass of 2'-Chloroacetophenone). During the distillation step, 2,4,6-trinitrophenol is employed as a polymerization inhibitor (inhibitor dosage: 25% by mass of o-chlorophenylethanol) to prevent intermolecular polymerization of o-chlorostyrene. [3]
Synthesis of cenobamate
Starting from 2'-Chloroacetophenone, the target compound cenobamate was synthesized through a sequence of α-bromination, condensation, and carbonyl reductase-mediated reduction to afford the chiral alcohol intermediate, followed by addition with chlorosulfonyl isocyanate and subsequent hydrolysis. As a key starting material, 2'-Chloroacetophenone served as the basis for the entire synthetic route. Results and conclusions: The structure of the target product was confirmed by ¹H-NMR, ¹³C-NMR, and ESI-MS spectra. The overall yield reached 38% (calculated based on 2'-Chloroacetophenone), with a purity exceeding 99.9% (determined by HPLC) and an ee value of 99.54%. This method exhibits advantages such as high stereoselectivity and environmental friendliness, providing a chemoenzymatic pathway for the industrial production of cenobamate.[4]
Reference
[1] Dong, Longlong; et al, Bidentate ligand-enabled Wacker-type oxidation of olefins using K2S2O8 as an oxidant, New Journal of Chemistry (2025), 49(43), 18886-18892.
[2] Xing, Dong ; et al, Selective Oxidation of Benzyl and Allyl Alcohols to Carbonyl Compounds or Acids Mediated by Iron Photocatalysts, ChemCatChem 2025, 17, e00611.
[3] Chen, Y. D. Optimization of Synthesis Process for o-Chlorostyrene[J]. Fine Chemical Intermediates, 2009, 39(1): 3.
[4] Huang, J. X.; Wang, H. Y.; Wu, Z. N.; et al. Study on the Synthetic Process of Cenobamate [J]. Chinese Journal of Medicinal Chemistry, 2021, 31(6): 4.
You may like
Lastest Price from 2'-Chloroacetophenone manufacturers

US $0.00/KG2025-04-21
- CAS:
- 2142-68-9
- Min. Order:
- 1KG
- Purity:
- 98%min
- Supply Ability:
- 30tons/month

US $10.00/KG2025-04-21
- CAS:
- 2142-68-9
- Min. Order:
- 1KG
- Purity:
- 99%
- Supply Ability:
- 10 mt